Advertisements
Advertisements
प्रश्न
Four elements P, Q, R and S belong to the third period of the Modern Periodic Table and have respectively 1, 3, 5 and 7 electrons in their outermost shells. Write the electronic configurations of Q and R and determine their valences. Write the molecular formula of the compound formed when P and S combine.
Advertisements
उत्तर
| Elements | Valence Electrons |
| P | 1 |
| Q | 3 |
| R | 5 |
| S | 7 |
Electronic configuration of Q = 2, 8, 3
If the number of electrons in the valence shell is less than 4, then the same number of electrons will represent the valency of that element.
Valency of Q = 3
Electronic configuration of R = 2, 8, 5
If the number of valence electrons is either equal to or greater than 4, then that number is subtracted from 8 to obtain the correct valency of that element.
Valency of R = 8 − 5 = 3
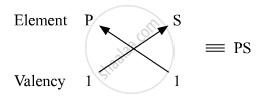
Molecular formula of the compound formed when P and S combine:
Number of valence electrons in P = 1
Valency of P = 1
Number of valence electrons in S = 7
Valency of S = 8 − 7 = 1