Advertisements
Advertisements
प्रश्न
State the following:
The group to which the element with an electronic configuration of 2, 8, 2 belongs.
Advertisements
उत्तर
Element [2, 8, 2] belongs to group 2 or [II – A]
APPEARS IN
संबंधित प्रश्न
Consider two elements 'A' (Atomic number 17) and 'B' (Atomic number 19) :
(i) Write the positions of these elements in the modern periodic table giving justification.
(ii) Write the formula of the compound formed when 'A' combines with 'B.'
(iii) Draw the electron dot structure of the compound and state the nature of the bond formed between the two elements.
An element with the atomic number 19 will most likely combine chemically with the element whose atomic number is ______.
Rewrite the following statement after correction, if necessary:
Elements in the same period have equal valency
What happens to the metallic character of the elements as we move from left to right in a period of the periodic table?
An element 'M' has atomic number 12.
(a) Write its electronic configuration.
(b) State the group to which 'M' belongs.
(c) Is 'M' a metal or a non-metal.
(d) Write the formula of its chloride.
Study the extract of the Periodic Table given below and answer the questions that follow. Give the alphabet corresponding to the element in question. DO NOT repeat an element. 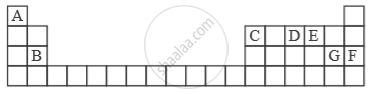
The ion of which element will migrate towards the cathode during electrolysis?
Study the extract of the Periodic Table given below and answer the questions that follow. Give the alphabet corresponding to the element in question. DO NOT repeat an element. 
Which is an inert gas?
Fill in the blank
There are ______ groups and ______ periods in the modern form of periodic table.
What is meant by a group in the periodic table?
Name and state the following with reference to the elements of the first three periods of the periodic table.
Non-metallic elements present in Period 3 of Groups 15 and 16.
