Advertisements
Advertisements
प्रश्न
Predict the major product, when 2-methyl-but-2-ene is converted into alcohol in the following method.
Hydroxylation using bayers reagent
Advertisements
उत्तर
\[\begin{array}{cc}
\phantom{.........................................}\ce{OH}\phantom{.}\ce{OH}\phantom{..}\\
\phantom{......................................}|\phantom{....}|\phantom{}\\
\ce{CH3 - C = CH - CH3 ->[Cold KMnO4/OH^Θ][(O)] 3CH3 - C - CH - CH3}\\
\phantom{..}|\phantom{.......................................}|\phantom{........}\\
\phantom{.}\ce{\underset{(2-methyl-but-2-ene)}{CH3}}\phantom{......................}\ce{\underset{(2-methyl-butan-2, 3-diol)}{CH3}}\phantom{..}
\end{array}\]
APPEARS IN
संबंधित प्रश्न
The reaction can be classified as
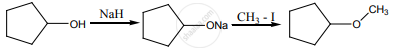
Arrange the following in the increasing order of their boiling point and give a reason for your ordering.
Propan-1-ol, propan-1, 2, 3-triol, propan-1, 3-diol, propan-2-ol
How is phenol prepared from isopropyl benzene?
Explain Kolbe’s reaction.
What will be the product (X and A)for the following reaction
acetylchloride \[\ce{{acetylchloride}->[i)CH3MgBr][ii)H3O+]X ->[acidK2Cr2O7] A}\]
What will be the product (X and A) for the following reaction:
\[\ce{acetylchloride ->[i) CH3MgBr][ii) H3O+]X->[acid K2Cr2O7]A}\]
What will be the product (X and A)for the following reaction
\[\ce{acetylchloride ->[i) CH3MgBr][ii) H3O] X ->[acid K2Cr2O7] A}\]
Identify the product(s) is/are formed when 1-methoxy propane is heated with excess HI. Name the mechanism involved in the reaction.
Predict the major product, when 2-methyl but -2-ene is converted into an alcohol in each of the following methods.
Acid catalysed hydration
Draw the major product formed when 1-ethoxyprop-1-ene is heated with one equivalent of HI.
