Advertisements
Advertisements
प्रश्न
Arrange the following in the increasing order of their boiling point and give a reason for your ordering.
Propan-1-ol, propan-1, 2, 3-triol, propan-1, 3-diol, propan-2-ol
Advertisements
उत्तर
2° alcohols have lower boiling points than 1° alcohol due to a corresponding decrease in the extent of H-bonding because of steric hindrance. Therefore Propan-1-ol has a higher boiling point than Propan-2-ol. Hydrogen group increases, boiling point also increases. Overall increasing order of boiling points is, propan-2-ol < Propan-1-ol < propan-1, 3-diol < propan-1, 2, 3-triol.
APPEARS IN
संबंधित प्रश्न
\[\ce{(CH3)3 - C - CH(OH) CH3 ->[con H2SO4] X (major product)}\]
The reaction is an example of
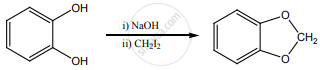
0.44 g of a monohydric alcohol, when added to methyl magnesium iodide in ether, liberates at STP 112 cm3 of methane with PCC the same alcohol form a carbonyl compound that answers the silver mirror test. Identify the compound.
What will be the product (X and A) for the following reaction?
\[\ce{acetylchloride ->[i) CH3MgBr][ii) H3O^+] X ->[acid K2Cr2O7] A}\]
Draw the major product formed when 1-ethoxyprop-1-ene is heated with one equivalent of HI
Identify the product (s) is / are formed when 1 – methoxy propane is heated with excess HI. Name the mechanism involved in the reaction
What will be the product (X and A) for the following reaction.
\[\ce{acetylchloride ->[i) CH3MgBr][ii) H3O^+] X ->[acid K2Cr2O7] A}\]
Identify the product(s) is/are formed when 1-methoxy propane is heated with excess HI. Name the mechanism involved in the reaction.
Predict the major product, when 2-methyl but -2-ene is converted into an alcohol in each of the following methods.
Acid catalysed hydration
The correct IUPAC name of the compound,
\[\begin{array}{cc}
\ce{CH3}\phantom{......}\\
|\phantom{........}\\
\ce{H3C - CH - CH - CH - CH2 - OH}\\
|\phantom{............}|\phantom{........}\\
\ce{Cl}\phantom{...........}\ce{CH3}\phantom{......}
\end{array}\]
