Advertisements
Advertisements
प्रश्न
Name the compound formed when ethanol is heated in excess of conc. sulphuric acid at 443 K. Also, write the chemical equation of the reaction stating the role of conc. sulphuric acid in it. What would happen if hydrogen is added to the product of this reaction in the presence of a catalyst such as palladium or nickel?
Advertisements
उत्तर
The compound that is formed when ethanol is heated in excess of conc. sulphuric acid at 443 K is ethene.
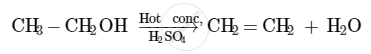
The concentrated sulphuric acid acts as a dehydrating agent in this reaction. It facilitates the removal of a molecule of water from the ethanol molecule.
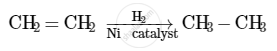
When hydrogen is added to ethene in the presence of a catalyst such as palladium or nickel, saturation of the compound takes place to form ethane.
APPEARS IN
संबंधित प्रश्न
What is the molecular formula and structure of the alcohol which can be thought to be derived from pentane?
Name one liquid carbon compound which is being used as an additive in petrol in some countries.
How would you convert of thanol into ethene?
One of the following substances is not added to make denatured alcohol. This is:
(a) methyl alcohol
(b) copper sulphate
(c) chloroform
(d) pyridine
How is the absolute alcohol obtained?
Name the addition product formed between ethene and water.
Give the balanced chemical equation of the following reaction:
Oxidation of ethanol by acidified potassium dichromate.
Give the balanced chemical equation of the following reaction:
Oxidation of ethanol by acidified potassium dichromate.
Give the balanced chemical equation of the following reaction:
Oxidation of ethanol by acidified potassium dichromate.
Give the balanced chemical equation of the following reaction:
Oxidation of ethanol by acidified potassium dichromate.
