Advertisements
Advertisements
प्रश्न
Name the compound formed when ethanol is heated in excess of conc. sulphuric acid at 443 K. Also, write the chemical equation of the reaction stating the role of conc. sulphuric acid in it. What would happen if hydrogen is added to the product of this reaction in the presence of a catalyst such as palladium or nickel?
Advertisements
उत्तर
The compound that is formed when ethanol is heated in excess of conc. sulphuric acid at 443 K is ethene.
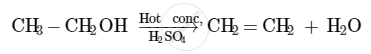
The concentrated sulphuric acid acts as a dehydrating agent in this reaction. It facilitates the removal of a molecule of water from the ethanol molecule.
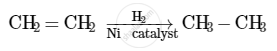
When hydrogen is added to ethene in the presence of a catalyst such as palladium or nickel, saturation of the compound takes place to form ethane.
APPEARS IN
संबंधित प्रश्न
How can ethanol and ethanoic acid be differentiated on the basis of their physical properties?
Write a balanced chemical equations for Heating of Ethanol at 170°C in the presence of conc. Sulphuric acid
Match the formulae in group A with appropriate names from group B:
Group A: CH3COOH, CH3CHO, CH3OH
Group B: Ethanol, Methanol, Ethanal, Ethanoic acid
What is meant by a functional group? Explain with an example.
Write the name and molecular formula of an organic compound having its name suffixed with 'ol' and having two carbon atoms in its molecule. Write balanced chemical equation to indicate what happens when this compound is heated with excess conc. H2SO4 and the name of main product formed. Also state the role of conc. H2SO4 in the reaction.
Find the odd one out and explain:
Why is the conversion of ethanol to ethanoic acid considered an oxidation reaction? Write the oxidising agent used in the reaction involved.
Draw the structures for ethanol.
Write the characteristics of ethanol.
Give the balanced chemical equation of the following reaction:
Oxidation of ethanol by acidified potassium dichromate.
