Advertisements
Advertisements
प्रश्न
Mention any three methods of preparation of haloalkanes from alcohols.
Describe various methods of preparation of haloalkanes from alcohols.
Advertisements
उत्तर
Haloalkanes are prepared by the following methods.
From alcohols: Alcohol can be converted into halo alkenes by reacting it with any one of the following reagents.
- Hydrogen halide
- Phosphorous halides
- Thionyl chloride.
1. Reaction with hydrogen halide:
\[\ce{\underset{\text{Ethanol}}{CH3CH2}OH + HCl ->[Anhydrous ZnCl2][\Delta] \underset{\text{Chloroethane}}{CH3CH2Cl + H2 O}}\]
Mixture of con. HCl and anhydrous ZnCl2 is called Lucas Reagent.
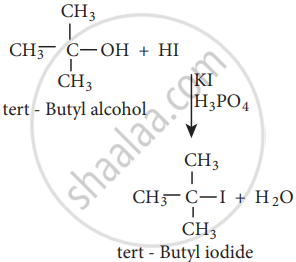
The order of reactivity of halo acids with alcohol is in the order HI > HBr > HCl.
The order of reactivity of alcohols with halo acid is tertiary > secondary > primary.
2. Reaction with phosphorous halides:
Alcohols react with PX5 or PX3 to form haloalkanes.
Example:
\[\ce{\underset{\text{Ethane}}{CH3CH2}OH + PCl5 ->\underset{\text{Chloro ethane}}{CH3CH2Cl +}POCl3 + HCl}\]
\[\ce{\underset{\text{Ethanol}}{3CH3CH2OH} + PCl3 -> \underset{\text{Chloro ethane}}{3 CH3CH2Cl +} H3PO3}\]
3. Reaction with Thionyl chloride(Sulphonyl Chloride)
\[\ce{\underset{\text{Ethanol}}{CH3CH3OH} + SOCl2 ->[Pyridine] \underset{\text{Chloro ethane}}{CH3CH2Cl +} SO2↑ + HCl↑}\]
APPEARS IN
संबंधित प्रश्न
Benzene reacts with Cl2 in the presence of FeCl3 and in absence of sunlight to form ______.
Match the compounds given in Column I with suitable items given in Column II.
| Column I (Compound) | Column II (Uses) |
||
| A | Iodoform | 1 | Fire extinguisher |
| B | Carbon tetra chloride | 2 | Insecticide |
| C | CFC | 3 | Antiseptic |
| D | DDT | 4 | Refrigerants |
Which one of the following is most reactive towards nucleophilic substitution reaction?
Give reasons for the polarity of C – X bond in haloalkane.
t – butyl chloride reacts with aqueous KOH by SN1 mechanism while n – butyl chloride reacts with SN2 mechanism.
p – dichlorobenzene has a higher melting point than those of o – and m – dichlorobenzene.
Write a short note on Dows Process.
Complete the following reaction.
\[\ce{CH3 - CH = CH2 + HBr ->[Peroxide]}\]
Complete the following reaction.
\[\ce{CH3 - CH2 - Br + NaSH ->[alcohol][H2O]}\]
The simplest alkene (A) reacts with HCl to form a compound (B). Compound (B) reacts with ammonia to form compound (C) of molecular formula C2H7N. Compound (C) undergoes carbylamine test. Identify (A),
(B) and (C).
