Advertisements
Advertisements
प्रश्न
Give reasons for the polarity of C – X bond in haloalkane.
थोडक्यात उत्तर
Advertisements
उत्तर
- Carbon halogen bond is a polar bond as halogens are more electronegative than carbon. The carbon atom exhibits a partial positive charge (δ+) and halogen atom a partial negative charge (δ–).
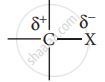
- The C -X bond is formed by the overlap of sp3 orbital of a carbon atom with the half-filled p- orbital of the halogen atom. The atomic size of halogen increases from fluorine to iodine, which increases the C – X bond length. Larger the size, greater is the bond length, and the weaker is the bond formed. The bond strength of C – X decreases from C – F to C – I in CH3X.
shaalaa.com
या प्रश्नात किंवा उत्तरात काही त्रुटी आहे का?
