Advertisements
Advertisements
प्रश्न
Mention any three methods of preparation of haloalkanes from alcohols.
Describe various methods of preparation of haloalkanes from alcohols.
Advertisements
उत्तर
Haloalkanes are prepared by the following methods.
From alcohols: Alcohol can be converted into halo alkenes by reacting it with any one of the following reagents.
- Hydrogen halide
- Phosphorous halides
- Thionyl chloride.
1. Reaction with hydrogen halide:
\[\ce{\underset{\text{Ethanol}}{CH3CH2}OH + HCl ->[Anhydrous ZnCl2][\Delta] \underset{\text{Chloroethane}}{CH3CH2Cl + H2 O}}\]
Mixture of con. HCl and anhydrous ZnCl2 is called Lucas Reagent.
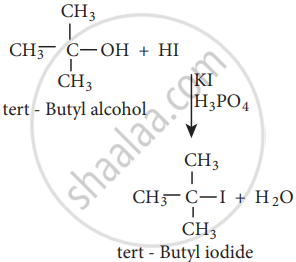
The order of reactivity of halo acids with alcohol is in the order HI > HBr > HCl.
The order of reactivity of alcohols with halo acid is tertiary > secondary > primary.
2. Reaction with phosphorous halides:
Alcohols react with PX5 or PX3 to form haloalkanes.
Example:
\[\ce{\underset{\text{Ethane}}{CH3CH2}OH + PCl5 ->\underset{\text{Chloro ethane}}{CH3CH2Cl +}POCl3 + HCl}\]
\[\ce{\underset{\text{Ethanol}}{3CH3CH2OH} + PCl3 -> \underset{\text{Chloro ethane}}{3 CH3CH2Cl +} H3PO3}\]
3. Reaction with Thionyl chloride(Sulphonyl Chloride)
\[\ce{\underset{\text{Ethanol}}{CH3CH3OH} + SOCl2 ->[Pyridine] \underset{\text{Chloro ethane}}{CH3CH2Cl +} SO2↑ + HCl↑}\]
