Advertisements
Advertisements
प्रश्न
Match the type of unit cell given in Column I with the features given in Column II.
| Column I | Column II |
| (i) Primitive cubic unit cell | (a) Each of the three perpendicular edges compulsorily have the different edge length i.e; a ≠ b ≠ c. |
| (ii) Body centred cubic unit cell | (b) Number of atoms per unit cell is one. |
| (iii) Face centred cubic unit cell | (c) Each of the three perpendicular edges compulsorily have the same edge length i.e; a = b = c. |
| (iv) End centred orthorhombic cell | (d) In addition to the contribution from unit cell the corner atoms the number of atoms present in a unit cell is one. |
| (e) In addition to the contribution from the corner atoms the number of atoms present in a unit cell is three. |
Advertisements
उत्तर
| Column I | Column II |
| (i) Primitive cubic unit cell | (b) Number of atoms per unit cell is one. |
| (c) Each of the three perpendicular edges compulsorily have the same edge length i.e; a = b = c. | |
| (ii) Body-centred cubic unit cell | (c) Each of the three perpendicular edges compulsorily have the same edge length i.e; a = b = c. |
| (d) In addition to the contribution from unit cell the corner atoms the number of atoms present in a unit cell is one. | |
| (iii) Face centred cubic unit cell | (c) Each of the three perpendicular edges compulsorily have the same edge length i.e; a = b = c. |
| (e) In addition to the contribution from the corner atoms the number of atoms present in a unit cell is three. | |
| (iv) End-centred orthorhombic cell | (a) Each of the three perpendicular edges compulsorily have the different edge length i.e; a ≠ b ≠ c. |
| (d) In addition to the contribution from unit cell the corner atoms the number of atoms present in a unit cell is one. |
Explanation:
(i) 
For primitive unit cell, a = b = c
Total number of atoms per unit cell = `1/8 xx 8` = 1
Here, `1/8` is due to contribution of each atom present at comer.
(ii)
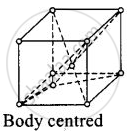
For body centered cubic unit cell, a = b = c.
This lattice contains atoms at comer as well as body centre.
Contribution due to atoms at comer = `1/8 xx 8` = 1
Contribution due to atoms at body centre = 8
(iii)
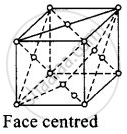
For face centred unit cell, a = b = c
Total constitutent ions per cell present at corners = `1/8 xx 8` = 1
Total constitutent ions per unit cell present at face centre = `1/2 xx 6` = 3
(iv)
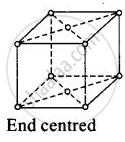
For end centred orthorhombic unit cell, a ≠ b ≠ c
Total contribution of atoms present at corner = `1/8 xx 8` = 1
Total constibution of atoms present at end centre = `1/2 xx 2` = 1
APPEARS IN
संबंधित प्रश्न
A unit cell of iron crystal has edge length 288 pm and density 7.86 g.cm-3. Find the number of atoms per unit cell and type of the crystal lattice.
Given : Molar mass of iron = 56 g.mol-1; Avogadro's number NA = 6.022 x 1023.
Face centred cubic crystal lattice of copper has density of 8.966 g.cm-3. Calculate the volume of the unit cell. Given molar mass of copper is 63.5 g mol-1 and Avogadro number NA is 6.022 x 1023 mol-1
An element crystallises in a b.c.c lattice with cell edge of 500 pm. The density of the element is 7.5g cm-3. How many atoms are present in 300 g of the element?
An element with molar mass 27 g mol−1 forms a cubic unit cell with edge length 4.05 ✕ 10−8 cm. If its density is 2.7 g cm−3, what is the nature of the cubic unit cell?
Explain how much portion of an atom located at (i) corner and (ii) body-centre of a cubic unit cell is part of its neighbouring unit cell.
How can you determine the atomic mass of an unknown metal if you know its density and the dimension of its unit cell? Explain.
Explain with reason sign conventions of ΔS in the following reaction
N2(g) + 3H2(g) → 2NH3(g)
A face centred cube (FCC) consists of how many atoms? Explain
An element 'X' (At. mass = 40 g mol-1) having f.c.c. the structure has unit cell edge length of 400 pm. Calculate the density of 'X' and the number of unit cells in 4 g of 'X'. (NA = 6.022 × 1023 mol-1)
Volume of unit cell occupied in face-centered cubic arrangement is ____________.
A metal has a body-centered cubic crystal structure. The density of the metal is 5.96 g/cm3. Find the volume of the unit cell if the atomic mass of metal is 50.
Gold has a face-centered cubic lattice with an edge length of the unit cube of 407 pm. Assuming the closest packing, the diameter of the gold atom is ____________.
The number of atoms contained in a fcc unit cell of a monoatomic substance is ____________.
Which of the following metal(s) show(s) hexagonal close-packed structure (hcp) and which show face-centered cubic (fcc) structure?
The density of a metal which crystallises in bcc lattice with unit cell edge length 300 pm and molar mass 50 g mol−1 will be:
The percentage of empty space in a body centred cubic arrangement is ______.
The coordination number for body center cubic (BCC) system is
If a represents the edge length of the cubic systems, i.e. simple cubic, body centred cubic and face centered cubic, then the ratio of the radii of the sphere in these system will be:-
A solid is formed by 2 elements P and Q. The element Q forms cubic close packing and atoms of P occupy one-third of tetrahedral voids. The formula of the compound is ______.
