Advertisements
Advertisements
प्रश्न
Identify the chiral molecule in the following pair :

Advertisements
उत्तर
Out of the given molecules, (i) is a chiral molecule since the C-atom (marked as * in the figure) is chiral and different groups are attached to it. This molecule does not have the centre of symmetry.
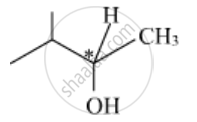
shaalaa.com
या प्रश्नात किंवा उत्तरात काही त्रुटी आहे का?
