Advertisements
Advertisements
प्रश्न
How will you prove that hydrochloric acid contains
- hydrogen
- chlorine?
Write equations for the reactions.
Advertisements
उत्तर
We can prove that hydrochloric acid contains both hydrogen and chlorine by the following experiment:
Take a voltameter used for water electrolysis with a platinum cathode and a graphite anode. Pour 4 molars of HCl into the voltameter and measure the direct electric current. The cathode emits a colourless gas, while the anode produces a greenish-yellow one. If you put a burned splinter close to a colourless gas, the gas will burst into flames, showing that it is hydrogen gas. Wet starch iodide paper turns blue-black when immersed in the gas, confirming its identity as chlorine.
Chemical equation: \[\ce{2HCl -> H2 ^ + Cl2 ^}\]
This experiment proves that hydrochloric acid contains both hydrogen and chlorine.
APPEARS IN
संबंधित प्रश्न
Name two gases which chemically combine to form a liquid.
Name the experiment which demonstrates that hydrogen chloride is soluble in water.
Complete and balance the following reaction, state whether dilute or conc. acid is used.
\[\ce{Pb(NO3)2 + HCl ->}\]
Complete the following reaction and balance them.
Zn+ 2HCl ⟶
Give a balanced equation when dilute hydrochloric acid is added to : Potassium bisulphite
Write the steps needed for flame test ?
A solution of hydrogen chloride in water is prepared. The following substances are added to separate portions of the solution:
| S. No. | Substances added | Gas evolved | Odour |
| 1. | Calcium carbonate | _________ | _________ |
| 2. | Magnesium ribbon | _________ | _________ |
| 3. | Manganese (IV) oxide with heating | _________ | _________ |
| 4. | Sodium sulphide | _________ | _________ |
Study the flow chart and give balanced equations with conditions for the conversions A, B, C, D and E.
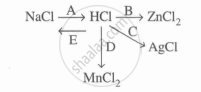
Complete and balance the following reaction, state whether dilute or cone. acid is used.
\[\ce{NH4OH + HCl->}\]
Complete and balance the following reaction, state whether dilute or conc. acid is used.
\[\ce{NH4OH + HCI->}\]
