Advertisements
Advertisements
प्रश्न
Name the gas evolved when dilute hydrochloric acid is added to: Potassium bisulphite
Advertisements
उत्तर
sulphur dioxide
APPEARS IN
संबंधित प्रश्न
Hydrochloric acid contains (i) hydrogen (ii) chlorine. Prove it. Write equations for the reactions.
Name a chloride which is solube in excess of ammonium hydroxide
Complete the following reaction and balance them.
Zn+ 2HCl ⟶
Name the following :
Acid present in the stomach of mammals.
Name the following:
Acid used to extract glue from bones.
Name the following: A greenish yellow gas.
Name the gas evolved when dilute hydrochloric acid is added to: Zinc Metal
Study the flow chart and give balanced equations with conditions for the conversions A, B, C, D and E.
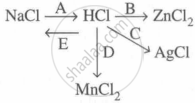
Complete and balance the following reaction, state whether dilute or conc. acid is used.
\[\ce{NH4OH + HCI->}\]
Complete and balance the following reaction, state whether dilute or cone. acid is used.
\[\ce{NH4OH + HCl->}\]
