Advertisements
Advertisements
प्रश्न
How can you obtain pure water from a salt-water mixture (or salt-solution) ? Draw a neat and labelled diagram of the apparatus you would use to obtain pure water from a salt-water mixture (or salt-solution).
Advertisements
उत्तर
We can obtain pure water from a saltwater mixture (or salt solution) by the process of distillation. Important steps underlying the process are as follows:
The salt water mixture is taken in a distillation flask and heated subsequently.
On heating, water vapours are formed that rise up and come through the side tube B.
It is then passed through condenser C.
Cold water from the tap is circulated in the outer tube of the condenser, so that the hot vapours are cooled.
The vapours condense and are collected in another beaker.
This pure water is called distilled water.
Salt, being non-volatile, remains in the flask and can be obtained later.
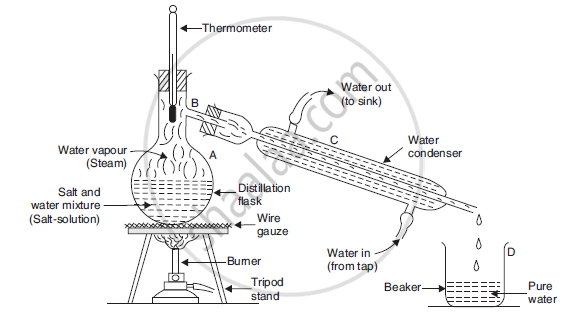
Separation by distillation. Here a mixture of salt and water (or salt-solution) is being separated by distillation.
APPEARS IN
संबंधित प्रश्न
What are the conditions for 'something' to be called 'matter'?
Describe in your own words, what happens to the particles when salt dissolves in water.
Name a metal which is soft and a non-metal which is hard.
Which type of elements, metals or non-metals, show the property of brittleness ?
What is the other name for impure substances ? Give two examples of impure substances.
Name a metal which can be easily cut with a knife.
Which of the following are compounds ?
- CO
- No
- NO
- Co
Which of the following is a sol ?
Shaving cream, Milk, Fog, Soap solution, Hairspray
You have been given a suspension and a solution. How could you tell the difference between them by their appearance ?
Name the different types of solutions. Give one example of each.
What difference in the property of two miscible liquids enables their separation by fractional distillation ?
Name the process you would use to separate a mixture of water and alcohol.
Which technique is used in a washing machine to squeeze out water from wet clothes while drying ?
Which technique can be used to detect and identify traces of poison present in the stomach wash of a person ?
How will you separate camphor, common salt and iron nails from their mixture ?
______ is not made of matter.
All the matter made of ______.
Stars including Sun are covered in ______.
Gases change to liquid by the process of ______.
Define the term matter.
