Advertisements
Advertisements
प्रश्न
How can you obtain pure water from a salt-water mixture (or salt-solution) ? Draw a neat and labelled diagram of the apparatus you would use to obtain pure water from a salt-water mixture (or salt-solution).
Advertisements
उत्तर
We can obtain pure water from a saltwater mixture (or salt solution) by the process of distillation. Important steps underlying the process are as follows:
The salt water mixture is taken in a distillation flask and heated subsequently.
On heating, water vapours are formed that rise up and come through the side tube B.
It is then passed through condenser C.
Cold water from the tap is circulated in the outer tube of the condenser, so that the hot vapours are cooled.
The vapours condense and are collected in another beaker.
This pure water is called distilled water.
Salt, being non-volatile, remains in the flask and can be obtained later.
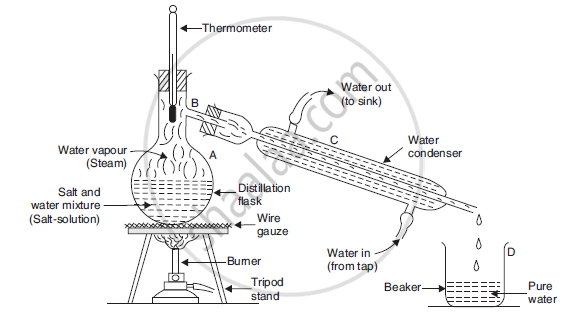
Separation by distillation. Here a mixture of salt and water (or salt-solution) is being separated by distillation.
APPEARS IN
संबंधित प्रश्न
Intermolecular force of attraction is maximum in ............ less in .......... and the least in .............
Name the terms for the particles of matter which may or may not have independent existence.
Bromine and air take about 15 minutes to diffuse completely but bromine diffuses into a vacuum very rapidly. Why is this so?
What is the general name of the materials which contain at least two pure substances and show the properties
of their constituents ?
Classify the following into elements and compounds :
- H2O
- He
- Cl2
- CO
- Co
What are pure substances ? Give two examples of pure substances.
Name two solid, two liquid and two gaseous elements at the room temperature.
Choose the correct answer.
A 5 per cent sugar solution means that :
Name the different types of solutions. Give one example of each.
What will happen if a saturated solution is :
- heated
- cooled
21.5 g of sodium chloride dissolves in 60 g of water at 25°C. Calculate the solubility of sodium chloride in water at that temperature.
Define solubility of a substance. How does it vary with temperature ?
What is a colloid ? Give two examples of colloids (or colloidal solutions)
Which of the following represents the solubility of sugar in water at 20°C ?
Draw a labelled diagram of the fractional distillation apparatus used for separating a mixture of alcohol and water.
The boiling point of liquid argon is :
Which among the following ______ will get attracted to by magnet?
You want to wear your favourite shirt to a party, but the problem is that it is still wet after a wash. What steps would you take to dry it faster?
The movement of particles in liquids is ______.
Define the term matter.
