Advertisements
Advertisements
प्रश्न
Give the structures and IUPAC names of monohydric phenols of molecular formula C7H8O.
रासायनिक समीकरणे/रचना
लघु उत्तर
Advertisements
उत्तर
The given molecular formula is C7H8O. As it is given that it is a phenol, the basic structure will be:

This structure accounts for C6H6O. When the remaining CH2 is added to phenol, it forms the compounds shown below.

2-Methylphenol (o-cresol)

3-Methylphenol (m-cresol)
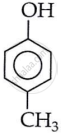
4-Methylphenol (p-cresol)
shaalaa.com
या प्रश्नात किंवा उत्तरात काही त्रुटी आहे का?
