Advertisements
Advertisements
प्रश्न
Give the chemical formula of ammonium sulphate
Advertisements
उत्तर
It denotes in a compound, the number of atoms of element present.
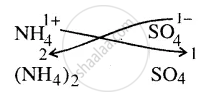
∴ Formula = \[\ce{(NH4)2SO4}\]
APPEARS IN
संबंधित प्रश्न
Fill in the following blank with suitable word:
The horizontal rows in a periodic table are called ...............
State whether the following statement is true or false:
On going down in a group of the periodic table, the number of valence electrons increases.
Name the noble gas with 3 shells.
Give the chemical formula of magnesium nitrate
What are groups and periods in the modern periodic table?
______ is the longest period in the periodical table.
What is the symbol of the element Tungsten?
“Hydrogen occupies a unique position in Modern Periodic Table”. Justify the statement.
An element has the atomic number 12.
- Write the electric configuration.
- To which group and period does it belong.
Give the criteria on the basis of which elements are placed (i) in a group and (ii) in a period in the modern periodic table. The atomic number of an element is 16. According to the criteria, in which group and period it should be placed?
