Advertisements
Advertisements
प्रश्न
Give reasons:Ferrimagnetic substances show better magnetism than antiferromagnetic substances.
Why do ferrimagnetic substances show better magnetism than antiferromagnetic substances?
Advertisements
उत्तर १
In the presence of magnetic field, the magnetic moments of domains of an antiferromagnetic substances are ordered in such a way half of the magnetic moments are aligned in one direction while the remaining half are in opposite direction. As a result of it net magnetic moment will be zero whereas in case of ferrimagnetic substances the magnetic moments of the domains of ferrimagnetic substances are aligned in parallel and anti-parallel directions in unequal numbers. As a result, there will be small value of magnetic moment for ferrimagnetic substances.
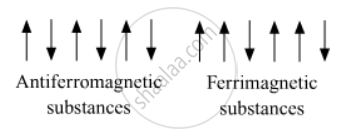
उत्तर २
Anti-ferromagnetism have domain structures that are oppositely oriented and cancel out each other's magnetic moment while in ferrimagnetism the magnetic moments of the domains in the substance are aligned in parallel and anti-parallel directions in unequal numbers resulting net dipole moment. Hence, ferrimagnetic substances show better magnetism than antiferromagnetic substances.
APPEARS IN
संबंधित प्रश्न
What type of magnetism is shown by a substance if magnetic moments of domains are arranged in same direction?
Analysis shows that nickel oxide has the formula Ni0.98O1.00. What fractions of nickel exist as Ni2+ and Ni3+ ions?
What is magnetic susceptibility?
Compare dia, para and ferromagnetism.
What happens to the domains in a ferromagnetic material in the presence of external magnetic field?
All those atoms or molecules which have an odd number of electrons are
Which one of the following would feel attraction when placed in magnetic field: Co2+, Ag+, Ti4+, Zn2+
Which one of the following homo-diatomic molecule is paramagnetic?
Some of the polar crystals when heated produces small electrical current. This phenomenon is called ______.
Which of the following is not paramagnetic?
