Advertisements
Advertisements
प्रश्न
Give reasons:Ferrimagnetic substances show better magnetism than antiferromagnetic substances.
Why do ferrimagnetic substances show better magnetism than antiferromagnetic substances?
Advertisements
उत्तर १
In the presence of magnetic field, the magnetic moments of domains of an antiferromagnetic substances are ordered in such a way half of the magnetic moments are aligned in one direction while the remaining half are in opposite direction. As a result of it net magnetic moment will be zero whereas in case of ferrimagnetic substances the magnetic moments of the domains of ferrimagnetic substances are aligned in parallel and anti-parallel directions in unequal numbers. As a result, there will be small value of magnetic moment for ferrimagnetic substances.
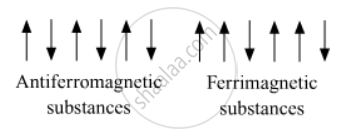
उत्तर २
Anti-ferromagnetism have domain structures that are oppositely oriented and cancel out each other's magnetic moment while in ferrimagnetism the magnetic moments of the domains in the substance are aligned in parallel and anti-parallel directions in unequal numbers resulting net dipole moment. Hence, ferrimagnetic substances show better magnetism than antiferromagnetic substances.
APPEARS IN
संबंधित प्रश्न
Iron (z=26) is highly ferromagnetic. Explain.
What type of magnetism is shown by a substance if magnetic moments of domains are arranged in same direction?
Write the type of magnetism observed when the magnetic moments are oppositely aligned and cancel out each other.
Explain the following with suitable examples:
Paramagnetism
Give reason for the following :
Sulphur in vapour state shows paramagnetic behaviour.
Compare the properties of soft and hard ferromagnetic materials.
Fe3O4 is ____________.
Fe3O4 (magnetite) is an example of ___________.
Which one of the following pairs has only paramagnetic species?
Magnetic materials used for making permanent magnets (P) and magnets in a transformer (T) have different properties of the following, which property best matches for the type of magnet required ?
