Advertisements
Advertisements
प्रश्न
Give its
(i) mathematical expression
(ii) graphical representation and
(iii) significance.
Advertisements
उत्तर
(i) mathematical expression
suppose a gas occupies volume V1 when its pressure is P1; then
`"V"_1 ∝ 1/"P"_1 "or" "V"_1 = "k"/"P"_1`
`"P"_1"V"_1 = "k" = "constant"`
If V2 is the volume ocuupied when the pressure is P2 at the same temperature, the
`"V"_2 ∝ 1/"P"_2 "or" "V"_2 = "k"/"P"_2`
`"P"_2"V"_2 = "k" = "constant"`
`"P"_1"V"_1 = "P"_2"V"_2 = "k"` at constant temperature.
This is called Boyle's law equation.
(ii) graphical representation
The law can be verified by plotting a graph
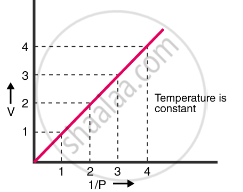
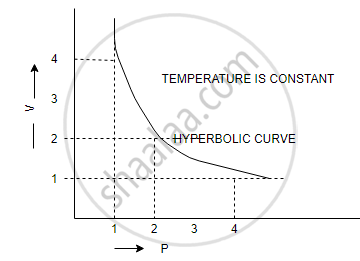
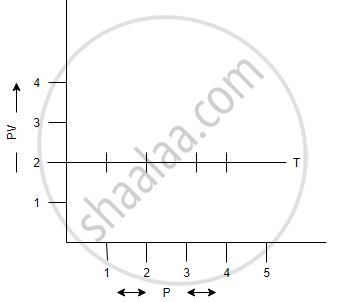
(a) V vs `1/p` (b) V vs P (c) PV vs P
(a) V vs `1/p`: a straight line passing through the origin is obtained
(b) V vs P: a hyperbolic curve in the first quadrant is obtained
(c) PV vs P: a straight line is obtained parallel to the pressure axis.
(iii) Significance
On increasing pressure, volume decreases. The gas becomes denser. Thus at a constant temperature, the density of a gas is directly proportional to its pressure.
Atmospheric pressure is low at high altitudes, so air is less dense. Hence, a lesser quantity of oxygen is available for breathing. This is the reason why mountaineers have to carry oxygen cylinders with them.
APPEARS IN
संबंधित प्रश्न
Explain Why?
Inflating a balloon seems to violate Boyle's law.
State the law which is represented by the following graph:

Choose the correct answer:
If the pressure is doubled for a fixed mass of a gas, its volume will become
A given mass of a gas occupied 143 cm3 at 27° C and 700 mm Hg pressure. What will be its volume at 300 K and 280 mm Hg pressure?
Calculate the volume of a gas ‘A’ at s.t.p., if at 37°C and 775 mm of mercury pressure, it occupies a volume of `9 1/2` litres.
Calculate the following:
A gas ‘X’ is collected over water at 17°C and 750 mm. pressure. If the volume of the gas collected is 50 cc., calculate the volume of the dry gas at s.t.p. [at 17°C the vapour pressure is 14 mm.]
Assuming temperature remaining constant calculate the pressure of the gas in the following:
The pressure of a gas having volume 100 lits. originally occupying 75 dm3 at 700 mm. pressure.
Assuming temperature remaining constant calculate the pressure of the gas in the following:
The pressure of a gas having volume 380 lits. originally occupying 800 cm3 at 76 cm. pressure.
Fill in the blank with the correct word, from the words in option:
1 dm3 of a gas is equal to _______.
According to Boyle’s law, the shape of the graph between pressure and reciprocal of volume is _______.
