Advertisements
Advertisements
प्रश्न
For the pair phenol and cyclohexanol, answer the following:
Why is phenol more acidic than cyclohexanol?
Advertisements
उत्तर
Because the conjugate base of phenol, the phenoxide ion, is more stable due to resonance, phenol is more acidic than cyclohexanol.

APPEARS IN
संबंधित प्रश्न
Give two reactions that show the acidic nature of phenol.
Explain how does the −OH group attached to a carbon of benzene ring activate it towards electrophilic substitution?
Account for the following:
o-nitrophenol is more steam volatile than p-nitrophenol.
Phenol is more acidic than alcohol because ____________.
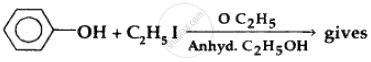
Which of the following compounds is most acidic?
Which one of the following compounds has the most acid nature?
What is the correct order of reactivity of alcohols in the following reaction?
\[\ce{R-OH + HCl ->[ZnCl2] R-Cl + H2O}\]
In the following compounds:
 |
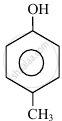 |
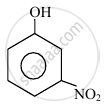 |
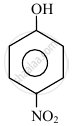 |
| (I) | (II) | (III) | (IV) |
The order to acidity is ______.
Give the structure of the product you would expect when the following alcohol reacts with HBr.
Butan-1-ol
