Advertisements
Advertisements
प्रश्न
Figure shows a cylindrical tube of cross-sectional area A fitted with two frictionless pistons. The pistons are connected to each other by a metallic wire. Initially, the temperature of the gas is T0 and its pressure is p0 which equals the atmospheric pressure. (a) What is the tension in the wire? (b) What will be the tension if the temperature is increased to 2T0 ?

Advertisements
उत्तर
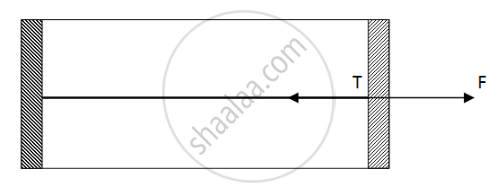
(a)
Since pressure from outside and inside the cylinder is the same, there is no net pressure acting on the pistons. So, tension will be zero.
(b)
`T_1 = T_0`
`T_2 = 2T_0`
`P_1 = P_0 = 10^5 "Pa"`
CSA = A
Let the Pistons be L distance apart .
V = AL
Applying five variable gas equation , we get
`(P_1V)/T_1 = (P_2V)/T_2`
⇒ `10^5/T_0 = P_2/(2T_0)`
⇒ `P_2 = 2×10^5 = 2P_0`
Net Force acting outside = `2P_0 - P_0 = P_0`
Force acting on a piston `F = P_0A`
By the free body diagram , we get
`F - T = 0`
`T = P_0A`
APPEARS IN
संबंधित प्रश्न
Can we define the temperature of (a) vacuum, (b) a single molecule?
A gas is kept in an enclosure. The pressure of the gas is reduced by pumping out some gas. Will the temperature of the gas decrease by Charles's low?
When you come out of a river after a dip, you feel cold. Explain.
The mean square speed of the molecules of a gas at absolute temperature T is proportional to
Which of the following quantities is the same for all ideal gases at the same temperature?
(a) The kinetic energy of 1 mole
(b) The kinetic energy of 1 g
(c) The number of molecules in 1 mole
(d) The number of molecules in 1 g
The temperature and the dew point in an open room are 20°C and 10°C. If the room temperature drops to 15°C, what will be the new dew point?
Answer in brief:
Show that rms velocity of an oxygen molecule is `sqrt2` times that of a sulfur dioxide molecule at S.T.P.
The emissive power of a sphere of area 0.02 m2 is 0.5 kcal s-1m-2. What is the amount of heat radiated by the spherical surface in 20 seconds?
Calculate the energy radiated in one minute by a blackbody of surface area 200 cm2 at 127 °C (σ = 5.7 x 10-8 J m-2 s-1 K-4)
What is the microscopic origin of temperature?
The average K.E. of hydrogen molecules at 27° C is E. The average K.E. at 627° C is ____________.
Volume versus temperature graphs for a given mass of an ideal gas are shown in figure at two different values of constant pressure. What can be inferred about relation between P1 and P2?

A gas mixture consists of molecules of types A, B and C with masses mA > mB > mC. Rank the three types of molecules in decreasing order of average K.E.
Consider a rectangular block of wood moving with a velocity v0 in a gas at temperature T and mass density ρ. Assume the velocity is along x-axis and the area of cross-section of the block perpendicular to v0 is A. Show that the drag force on the block is `4ρAv_0 sqrt((KT)/m)`, where m is the mass of the gas molecule.
For a particle moving in vertical circle, the total energy at different positions along the path ______.
Show that the average energy per molecule is directly proportional to the absolute temperature ‘T’ of the gas.
2000 calories of radiant heat is incident on a body. If the body absorbs 550 calories of heat, find the coefficient of emmission of the body.
