Advertisements
Advertisements
प्रश्न
Explain with reasons and diagrams, the positive and negative deviations from Raoult’s law shown by nonideal solutions.
Advertisements
उत्तर
- Positive deviation from Raoult’s law:
The solutions in which solute-solvent intermolecular attractions are weaker than those between solute-solute molecules and solvent-solvent molecules, exhibit positive deviations. The vapour pressures of such solutions are higher than those of pure components as shown in the diagram.
e.g. The solutions of ethanol + acetone, carbon disulphide + acetone show positive deviations from Raoult’s law.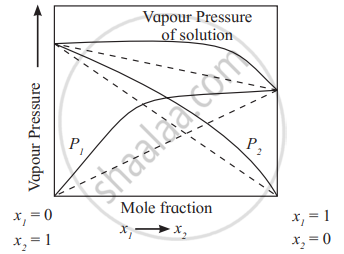
Positive derivations from Raoult's law - Negative deviation from Raoult’s law:
The solutions in which the interactions between solvents and solute molecules are stronger than solute-solute or solvent-solvent interactions, exhibit negative deviations. The vapour pressures of such solutions are lower than those of pure components as shown in the diagram.
e.g. Solutions of phenol + aniline, chloroform + acetone exhibit negative deviations from Raoult’s law.
Negative derivations from Raoult's law
APPEARS IN
संबंधित प्रश्न
Choose the most correct option.
The vapour pressure of a solution containing 2 moles of a solute in 2 moles of water (vapour pressure of pure water = 24 mm Hg) is ______.
Choose the most correct option.
Vapour pressure of a solution is _______.
State Raoult’s law for the solutions of non-volatile solutes in volatile solvents.
Answer the following.
Using Raoult’s law, how will you show that ΔP = `"P"_1^0 "x"_2`? Where, x2 is the mole fraction of solute in the solution and `"P"_1^0` vapour pressure of pure solvent.
A mixture of benzene and toluene contains 30% by mass of toluene. At 30°C, vapour pressure of pure toluene is 36.7 mm Hg and that of pure benzene is 118.2 mm Hg. Assuming that the two liquids form ideal solutions, calculate the total pressure and partial pressure of each constituent above the solution at 30°C.
State Raoult’s law.
What type of solutions exhibit positive deviations from Raoult’s law?
With the help of vapour pressure-temperature curves for solution and solvent, explain why boiling point of solvent is elevated when a nonvolatile solute is dissolved into it.
The vapour pressure of a pure solvent at a certain temperature is 0.0227 bar. What is the vapour pressure of a solution containing 6 g of solute (M = 60 g/mol) in 50 g of solvent?
What are non-ideal solutions?
Which of the following statements is INCORRECT?
Which of the following metals is refined by vapour phase refining in Mond process?
9 gram anhydrous oxalic acid (mol. wt. = 90) was dissolved in 9.9 moles of water. If vapour pressure of pure water is pf the vapour pressure of solution is ______.
5.0 g of sodium hydroxide (molar mass 40 g mol-1) is dissolved in little quantity of water and the solution is diluted upto 100 mL. What is the molarity of the resulting solution?
A solution of acetone in ethanol ______.
18 g of glucose (C6H12O6) is added to 178.2 g of water. The vapour pressure of water for this aqueous solution at l00°C is ______.
A solution having highest vapour pressure is ______.
Explain the variation of vapour pressures with mole fractions of binary solutions of two volatile liquids forming ideal solutions.
A mixture of two liquids A and B have vapour pressures 3.4 × 104 Mn-2 and 5.2 × 104 Nm-2. If the mole fraction of A is 0.85, find the vapour pressure of the solution.
The solutions that obey Raoult's law over entire range of concentrations are ______.
Which one of the following is not correct for an ideal solution?
