Advertisements
Advertisements
प्रश्न
Explain the use of radioactive in the field of medicine, agriculture and industry.
Advertisements
उत्तर
Uses of radioactivity in medical, agriculture and industry:
Uses in medical field:
(i) Rays from Radium produce satisfactorily improvement in skin diseases.
(ii) Radiation from Co60 is used in cancer treatment.
(iii) Radio I131 is used to diagnose and treat thyroid disorders.
Uses in agriculture:
(i) Radioactive Phosphorus P32 is used in the study of metabolism.
(ii) Radioactive Sulphur S35 helps to study advantages and disadvantages
of fungicides.
(iii) Pests and insects can be killed by Y-radiation.
Uses in industry:
(i) In manutacturing papers, plastic ·and m·etal sheets to control the
thickness of the sheets.
(ii) Radioisotopes can be used to estimate the amount of wear in bearings.
(iii) Radioisotopes can be used to detect crack in welding, casting etc.
APPEARS IN
संबंधित प्रश्न

Complete the diagram as given above by drawing the deflection of radioactive radiations in an electric field
What do you mean by Mass number.
State two similarities and two dissimilarities between the γ-rays and X-rays.
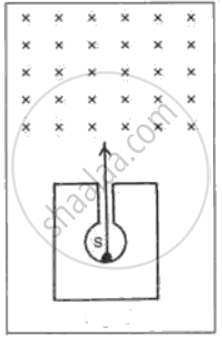
In following fig. shows a mixed source S of alpha and beta particles in a thick lead container. The particles pass through a magnetic field in a direction perpendicular to the plane of paper (inwards as shown by x). State and show in the diagram how the particles get affected.
Define free electrons.
What are the types of emission?
In a cathode ray tube state. How is it possible to increase the rate of emission of electrons.
How is a cathode ray tube used to convert an electrical signal into a visual signal?
State one use of radio-isotopes.
Which of the following statements is/are correct?
- α particles are photons
- Penetrating power of γ radiation is very low
- Ionization power is maximum for α rays
- Penetrating power of γ radiation is very high
