Advertisements
Advertisements
प्रश्न
Explain the concept of the photoelectric effect.
Advertisements
उत्तर
- The phenomenon of emission of electrons from a metal surface, when radiation of appropriate frequency is incident on it, is known as the photoelectric effect.
- The emitted electrons are called photoelectrons and the resulting current in the circuit due to them is called photoelectric current.
- When ultraviolet radiations fall on the emitter plate, electrons are ejected from it.
- They are attracted towards the positive collector plate by the electric field. Thus, light falling on the surface of the emitter causes a current in the external circuit.
APPEARS IN
संबंधित प्रश्न
Choose the correct option.
Polychromatic (containing many different frequencies) radiation is used in an experiment on the photoelectric effect. The stopping potential ______.
It is observed in an experiment on the photoelectric effect that an increase in the intensity of the incident radiation does not change the maximum kinetic energy of the electrons. Where does the extra energy of the incident radiation go? Is it lost? State your answer with explanatory reasoning.
The electrons are emitted in the photoelectric effect from a metal surface.
The maximum kinetic energy of the photoelectrons depends only on ______
Find the kinetic energy of the emitted electron, if in photoelectric effect energy of incident Photon is 4 eV and work function is 2.4 eV.
State Einstein photoelectric equation.
With the help of a circuit diagram describing an experiment to study the photoelectric effect.
The ratio of energies of photons produced due to transition of electron of hydrogen atom from its (i) second to first energy level and (ii) highest energy level to second level is respectively.
Which one of the following is TRUE in photoelectric emission?
When a photon enters glass from air, which one of the following quantity does not change?
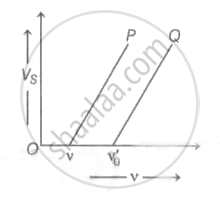
The graph of stopping potential `"V"_"s"` against frequency v of incident radiation is plotted for two different metals P and Q as shown in the graph. ΦP and ΦQ are work-functions of P and Q respectively, then
The work function of a metal is 1.6 x 10-19 J. When the metal surface is illuminated by the light of wavelength 6400 Å, then the maximum kinetic energy of emitted photo-electrons will be (Planck's constant h = 6.4 x 10-34 Js) ____________.
Light of wavelength `lambda` strikes a photo-sensitive surface and electrons are ejected with kinetic energy E. If the kinetic energy is to be increased to 2E, the wavelength must be changed to `lambda'` where ____________.
Threshold wavelength for lithium metal is 6250 Å. For photoemission, the wavelength of the incident light must be ______.
When light of wavelength 'λ' is incident on a photosensitive surface, the stopping potential is 'V'. When light of wavelength '3λ' is incident on the same surface, the stopping potential is `"V"/6`. Threshold wavelength for the surface is _______.
The photo electric effect to take place for a metal, the minimum frequency required is 5.792 × 1014 Hz. A light of wavelength 6000 Å is incident on that metal surface. What is the corresponding frequency of light and will there be photoelectric emissions? [velocity of light = 3 × 108 m/s]
The work function of a substance is 4.0 eV. The longest wavelength of light that can cause photo-emission from this substance is approximately (h = 6.63 × 10-34 Js)[1eV = 1.6 × 10-19 J]
The photon of frequency vis incident on a metal surface whose threshold frequency is v0. The kinetic energy of the emitted photoelectrons will be ____________.
When a photosensitive surface is irradiated by lights of wavelengths `lambda_1` and `lambda_2`, kinetic energies of emitted photoelectrons are E1 and E2 respectively. The work function of the photosensitive surface is ____________.
Photoelectrons are observed to just emit out of a material surface when the light of 620 nm falls on it with the intensity of 100 W m-2. If the light of wavelength 400 nm is incident on the same material with an intensity of 1 W m-2, what would be the minimum reverse potential needed to stop the outflow of the electrons?
The radiation emitted, when an electron jumps from n = 3 to n = 2 orbit is a hydrogen atom, falls on a metal to produce photoelectron. The electrons from the metal surface with maximum kinetic energy are made to move perpendicular to a magnetic field of `1/320`T in a radius of 10-3m. Find the 320 work function of metal:
On a photosensitive material when frequency of incident radiation is increased by 30%, kinetic energy of emitted photoelectrons increases from 0.4 eV. The work function of the surface is ______.
The following graphs show the variation of stopping potential corresponding to the frequency of incident radiation (ν) for a given metal. The correct variation is shown in graph [ν0 = threshold frequency].
|
(A) |
(B) |
|
(C) |
(D) |
The photoelectric threshold for a certain metal surface is 3600 Å. If the metal surface is irradiated by a wavelength of 1100 Å, then kinetic energy of the emitted photoelectrons is ______.
Define photoelectric work function of a metal.
By increasing the voltage in an electron diffraction tube, the radius of the diffraction rings will ______.
According to Einstein's photoelectric equation, the graph of kinetic energy of the photoelectron emitted from the metal versus the frequency of incident radiation gives a straight line graph whose slopе ______.




