Advertisements
Advertisements
प्रश्न
Explain the term of Metallurgy .
Advertisements
उत्तर
. Metallurgy: The various processes involved in the extraction of metals from its ores and refining is called metallurgy.
The major steps involved for the extraction of a metal from its ore are:
(i) Concentration of ores (or enrichment of ore)
(ii) Conversion of concentrated ore into metal
(iii) Refining (purification) of impure metal
APPEARS IN
संबंधित प्रश्न
Metal A has an electronic configuration of 2, 8, 1 and metal B has 2, 8, 8, 2 which is a more reactive metal.
Give the effect of heat on their: carbonates
Explain the following terms:
(a) flux
Explain the following terms:
(c) slag
`2Cu_2O +Cu_2S` ......................
......................
`HgS + O_2`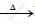 ..................
..................
what does it form with silica present in the ore?
Which metal is used for:
making pipes, buckets, water tanks,
What is magnalium? Name the main elements present in it? Write its one use ?
Aluminium is extracted from its chief ore, bauxite. The ore is first purified and then the metal is extracted from it by electrolytic reduction.
Write three balanced equations for the purification of bauxite.
Explain with reason:
Why is powdered coke sprinkled on top of the electrolyte?
Name the following:
Name an allotrope of a non-metal that allows electricity to pass through it.
The following is a sketch of an electrolytic cell used in the extraction of aluminium :
(a) What is the substance of which the electrode A and B are made?
(b) At which electrode (A or B) is aluminium formed?
(c) What are the two aluminium compounds in the electrolyte C?
(d) Why is it necessary for electrode B to be continuously replaced?
Write scientific reason.
Magnetic separation method is used to separate the magnetic ingredients in the ores.
Explain concept with example/explain with the help of a balanced equation.
Gangue
Identify the following method of concentration of ores and explain briefly.
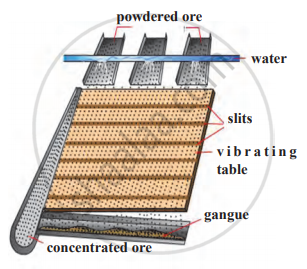
Observe the figure and name and explain in brief the following method:

State the reason for the addition of caustic alkali to bauxite ore during the purification of bauxite.
Explain the smelting process.
