Advertisements
Advertisements
प्रश्न
Ethylidene chloride is a/an ______.
पर्याय
vic-dihalide
gem-dihalide
allylic halide
vinylic halide
Advertisements
उत्तर
Ethylidene chloride is a/an gem-dihalide.
Explanation:
gem-dihalides are compounds in which both the halogen atoms are linked with the same carbon atom. They known as alkylidene halides \[\ce{CH3 - CHCl2}\] also. The structure of 'Ethylidene chloride' shows that both halogen atoms are present on same carbon atom.
APPEARS IN
संबंधित प्रश्न
Classify the following compound as a primary, secondary and tertiary halide.
2-Bromo-2-methylpropane
Match the structures of compounds given in Column I with the classes of compounds given in Column II.
| Column I | Column II | |
| (i) | \[\begin{array}{cc} \ce{CH3 - CH - CH3}\\ |\phantom{..}\\ \ce{X}\phantom{..} \end{array}\] |
(a) Aryl halide |
| (ii) | \[\ce{CH2 = CH - CH2 - X}\] | (b) Alkyl halide |
| (iii) | 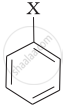 |
(c) Vinyl halide |
| (iv) | \[\ce{CH2 = CH - X}\] | (d) Allyl halide |
Which of the following belongs to the class of vinyl halides?
Name the following halides according to the IUPAC system and classify them as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halides:
CH3C(C2H5 )2CH2Br
Name the following halide according to IUPAC system and classify them as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halide:
\[\ce{CH3C(C2H5)2CH2Br}\]
Name the following halide according to the IUPAC system and classify it as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halide:
CH3C(Cl)(C2H5)CH2CH3
Name the following halide according to the IUPAC system and classify it as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halide:
\[\ce{CH_3CH_2C(CH_3)_2CH_2I}\]
Name the following halide according to IUPAC system and classify them as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halides:
\[\ce{CH3C(C2H5)2CH2Br}\]
Name the following halide according to IUPAC system and classify them as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halides:
\[\ce{CH3CH2C(CH3)2CH2I}\]
Write the structure of the following organic halogen compound.
1,4-Dibromobut-2-ene
Name the following halide according to IUPAC system and classify them as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halides:
\[\ce{CH3C(Cl)(C2H5)CH2CH3}\]
Name the following halide according to IUPAC system and classify it as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halides:
\[\ce{CH3C(Cl)(C2H5)CH2CH3}\]
Name the following halides according to IUPAC system and classify them as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halides:
\[\ce{CH3CH2C(CH3)2CH2I}\]
Name the following halides according to IUPAC system and classify them as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halides:
\[\ce{CH3C(C2H5)2CH2Br}\]
Give the IUPAC name of the compound.
\[\ce{CICH2 C ≡ CCH2 Br}\]
Name the following halide according to IUPAC system and classify as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halides:
\[\ce{CH3C(C2H5)2CH2Br}\]
Name the following halides according to IUPAC system and classify them as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halides:
\[\ce{CH3C(Cl)(C2H5)CH2CH3}\]
Name the following halide according to IUPAC system and classify as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halide:
\[\ce{CH3CH2C(CH3)2CH2I}\]
Which of the following is an example of vicdihalide?
Ethylidene chloride is a/an ______.
