Advertisements
Advertisements
प्रश्न
Draw the structure of major monohalo products in the following reaction:

Advertisements
उत्तर १
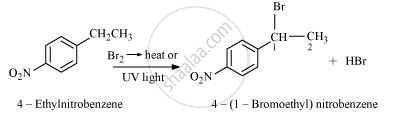
उत्तर २
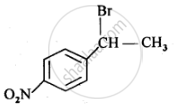
APPEARS IN
संबंधित प्रश्न
Name the following halide according to the IUPAC system and classify it as an alkyl, allyl, benzoyl (primary, secondary, tertiary), vinyl or aryl halide:
(CH3)2CHCH(Cl)CH3
Name the following halide according to the IUPAC system and classify it as an alkyl, allyl, benzoyl (primary, secondary, tertiary), vinyl or aryl halide:
CH3CH(CH3)CH(Br)CH3
Name the following halide according to the IUPAC system and classify it as an alkyl, allyl, benzoyl (primary, secondary, tertiary), vinyl or aryl halide:
p-ClC6H4CH2CH(CH3)2
Name the following halide according to the IUPAC system and classify it as an alkyl, allyl, benzoyl (primary, secondary, tertiary), vinyl or aryl halide:
o-Br-C6H4CH(CH3)CH2CH3
When two halogen atoms are attached to the same carbon atom then it is:
In which of the following molecules carbon atom marked with asterisk (*) is asymmetric?
(a) \[\begin{array}{cc}
\ce{H}\\
|\\
\phantom{.}\ce{C^*}\phantom{}\\
\phantom{}/\phantom{..}|\phantom{..}\backslash\phantom{}\\
\phantom{.}\ce{I}\phantom{...}\ce{Br}\phantom{..}\ce{Cl}
\end{array}\]
(b) \[\begin{array}{cc}
\ce{D}\\
|\\
\phantom{.}\ce{C^*}\phantom{}\\
\phantom{}/\phantom{..}|\phantom{..}\backslash\phantom{}\\
\phantom{.}\ce{I}\phantom{...}\ce{Br}\phantom{..}\ce{Cl}
\end{array}\]
(c) \[\begin{array}{cc}
\ce{H}\\
|\\
\phantom{.}\ce{C^*}\phantom{}\\
\phantom{}/\phantom{..}|\phantom{..}\backslash\phantom{}\\
\phantom{..}\ce{OH}\phantom{..}\ce{C2H5}\phantom{}\ce{CH3}
\end{array}\]
(d) \[\begin{array}{cc}
\ce{H}\\
|\\
\phantom{.}\ce{C^*}\phantom{}\\
\phantom{}/\phantom{..}|\phantom{..}\backslash\phantom{}\\
\phantom{..}\ce{H}\phantom{...}\ce{C2H5}\phantom{}\ce{CH3}
\end{array}\]
The position of \[\ce{-Br}\] in the compound in \[\ce{CH3CH = CH(Br)(CH3)2}\] can be classified as ______.
Name the following halide according to IUPAC system and classify them as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halide:
\[\ce{CH3C(C2H5)2CH2Br}\]
Name the following halide according to the IUPAC system and classify it as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halide:
\[\ce{CH_3CH_2C(CH_3)_2CH_2I}\]
Name the following halide according to IUPAC system and classify them as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halides:
\[\ce{CH3C(C2H5)2CH2Br}\]
Write the structure of the following organic halogen compound.
1,4-Dibromobut-2-ene
Name the following halide according to IUPAC system and classify them as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halides:
\[\ce{CH3C(C2H5)2CH2Br}\]
Name the following halide according to IUPAC system and classify as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halide:
\[\ce{CH3C(C2H5)2CH2Br}\]
Name the following halides according to IUPAC system and classify them as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halides:
\[\ce{CH3C(C2H5)2CH2Br}\]
Give the IUPAC name of the compound.
\[\ce{CICH2 C ≡ CCH2 Br}\]
Name the following halide according to IUPAC system and classify them as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halide:
\[\ce{CH3C(CI)(C2H5)CH2CH3}\]
Name the following halides according to IUPAC system and classify them as alkyl, allyl, benzyl (primary, secondary, tertiary), vinyl or aryl halides:
\[\ce{CH3C(Cl)(C2H5)CH2CH3}\]
Write the structure of the following organic halogen compound.
4-tert-Butyl-3-iodoheptane
What should be the correct IUPAC name for diethylbromomethane?
