Advertisements
Advertisements
प्रश्न
Draw the major product formed when 1-ethoxyprop-1-ene is heated with one equivalent of HI.
Advertisements
उत्तर
\[\begin{array}{cc}
\phantom{...............................................}\ce{H}\\
\phantom{...............................................}|\\
\ce{\underset{(1-ethoxy-prop-1-ene)}{CH3 - CH = CH} - \underset{\bullet\bullet}{\overset{\bullet\bullet}{O}} - C2H5 ->[H - I][\Delta] CH3 - CH = CH - O - C2H5}
\end{array}\]
\[\ce{\underset{(1-iodo-prop-1-ene)}{CH3 - CH = CH - I} <-[+I^Θ] CH3 - CH = \overset{⊕}{C}H + C2H5OH}\]
This reaction follows SN1 mechanism because in this reaction the more stable carbocation is formed that is double bonded carbocation. Therefore, the given molecule reacts with HI to form ethanol and 1-iodo prop-1-ene.
\[\ce{CH3 - CH = CH - O - C2H5 ->[HI/\Delta] \underset{(1-iodo-prop-1-ene)}{CH3 - CH = CH - I} + \underset{(ethanol)}{C2H5OH}}\]
APPEARS IN
संबंधित प्रश्न
 on treatment with Con. H2SO4 predominately gives
on treatment with Con. H2SO4 predominately gives
Suggest a suitable reagent to prepare secondary alcohol with the identical group using a Grignard reagent.
Predict the major product, when 2-methyl-but-2-ene is converted into alcohol in the following method.
Hydroboration
Explain Kolbe’s reaction.
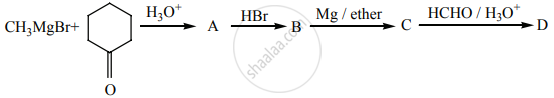
Identify A, B, C, D and write the complete equation.
What will be the product (X and A) for the following reaction:
\[\ce{acetylchloride ->[i) CH3MgBr][ii) H3O+]X->[acid K2Cr2O7]A}\]
Draw the major product formed when 1-ethoxyprop-1-ene is heated with one equivalent of HI.
What will be the product (X and A) for the following reaction.
\[\ce{acetylchloride ->[i) CH3MgBr][ii) H3O^+] X ->[acid K2Cr2O7] A}\]
Predict the major product, when 2-methyl but -2-ene is converted into an alcohol in each of the following methods.
Acid catalysed hydration
Draw the major product formed when 1-ethoxyprop-1-ene is heated with one equivalent of HI
