Advertisements
Advertisements
प्रश्न
Identify the product(s) is/are formed when 1-methoxy propane is heated with excess HI. Name the mechanism involved in the reaction.
Advertisements
उत्तर
\[\ce{\underset{1-methoxy propane}{CH3 - O - CH2 - CH2 - CH3} + HI -> \underset{\underset{(Iodomethane)}{Methyl Iodide}}{CH3I} + \underset{1-propane}{CH3 - CH2 - CH2 - OH} ->[HI] \underset{1-Iodopropane}{CH3 - CH2 - CH2 - I} + H2O}\]
Ethers having the primary alkyl group undergo an SN2 reaction.
APPEARS IN
संबंधित प्रश्न
Which one of the following will react with phenol to give salicylaldehyde after hydrolysis.
\[\ce{(CH3)3 - C - CH(OH) CH3 ->[con H2SO4] X (major product)}\]
In the reaction \[\ce{Ethanol ->[PCl5] X ->[alc. KOH] Y ->[H2SO4/H2O][298 K] Z}\]. The ‘Z’ is
HO CH2 CH2 – OH on heating with periodic acid gives ____________.
How is the conversion effected benzyl chloride to benzyl alcohol?
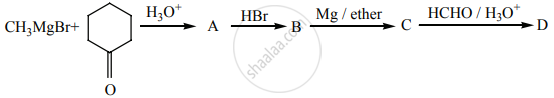
Identify A, B, C, D and write the complete equation.
What will be the product (X and A) for the following reaction?
acetylchloride \[\ce{->[i) CH3MgBr][ii) H3O+] X ->[acid K2Cr2O7] A}\]
Draw the major product formed when 1-ethoxyprop-1-ene is heated with one equivalent of HI.
What will be the product (X and A) for the following reaction
\[\ce{acetylchloride ->[i) CH3MgBr][ii) H3O^+ ]X ->[acid K2Cr2O7]A}\]
What will be the product (X and A) for the following reaction?
acetylchloride\[\ce{->[i) CH3MgBr][ii) H3O^+]X->[acid K2Cr2O7]A}\]
