Advertisements
Advertisements
प्रश्न
Draw structure of Chlorine trifluoride
Advertisements
उत्तर
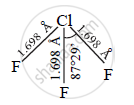
Structure of Chlorine trifluoride
APPEARS IN
संबंधित प्रश्न
Draw the structures of the following molecules: BrF3
ICl is more reactive than I2.
Draw the structure of BrF5
Draw the structures of the following : ClF3
Why is ICl more reactive than I2?
Draw the structures of the following: BrF5
Draw the structure of IF7. Write its geometry and the type of hybridization.
Explain why:
On being slowly passed through water, PH3 forms bubbles but NH3 dissolves.
Find the INCORRECT match.
Which among the following pairs of halogen forms the interhalogen compound of the type \[\ce{XX^{'}_7}\]?
What is the oxidation state of bromine in the product?
\[\ce{Br2 + \underset{(excess)}{3F2} ->?}\]
The CORRECT statement about bromine trifluoride is:
Which of the following exists in a yellow powder form at 25°C?
Which of the following interhalogen is a pale brown gas at room temperature?
The shape of BrF5 molecule is ______.
Which of the following interhalogen is a colourless liquid at room temperature?
BrF undergoes disproportionation to form Br2 and another interhalogen compound. What is the oxidation state of Br in this interhalogen compound?
Which among the following halogen does not form polyhalide ion?
What type of hybridization is observed in interhalogen compounds of the type \[\ce{XX^'_3}\]?
In the interhalogen compound AB3, the state of hydridisation of A is ______.
The interhalogen compound fonned from the reaction of bromine with excess of fluorine is a ______.
Brown ring test is used for detection of which radical?
Which of the following interhalogen compounds is used in the determination of iodine value of an oil?
