Advertisements
Advertisements
प्रश्न
Draw structure of Chlorine trifluoride
Advertisements
उत्तर
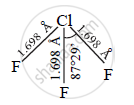
Structure of Chlorine trifluoride
APPEARS IN
संबंधित प्रश्न
Explain the structure of ClF3
Account for the following: Interhalogens are more reactive than pure halogens.
Draw the structure of BrF5
Draw the structures of the following : ClF3
Why is ICl more reactive than I2?
Draw the structures of the following: BrF5
Draw the structure of IF7. Write its geometry and the type of hybridization.
Explain why:
On being slowly passed through water, PH3 forms bubbles but NH3 dissolves.
Find the INCORRECT match.
Which among the following pairs of halogen forms the interhalogen compound of the type \[\ce{XX^{'}_7}\]?
The CORRECT statement about bromine trifluoride is:
Which among the following does not form polyhalide ion?
Which among the following is the most reactive?
Which of the following interhalogen is a pale brown gas at room temperature?
The shape of BrF5 molecule is ______.
Which of the following interhalogen is a colourless liquid at room temperature?
The number of lone pair of electrons present on central atom in the interhalogen XX'3 molecule:
What type of hybridization is observed in interhalogen compounds of the type \[\ce{XX^'_3}\]?
Which of the following is not the characteristic of interhalogen compounds?
The interhalogen compound fonned from the reaction of bromine with excess of fluorine is a ______.
Brown ring test is used for detection of which radical?
