Advertisements
Advertisements
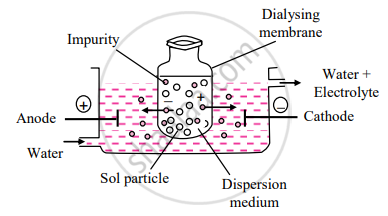
प्रश्न
Draw labelled diagram Dialysis.
Advertisements
उत्तर
APPEARS IN
संबंधित प्रश्न
Write one difference in Multimolecular colloid and Associated colloid
Define the following with a suitable example, of each:
Multimolecular colloid
Define the term Electrophoresis.
Explain Electrophoresis diagramatically.
Which type of colloid is formed on the dissolution of soap in water?
In coagulation of positively charged ferric hydroxide sol, the most and least effective ions are ____________ and ____________ respectively.
Which of the following is a heterogeneous mixture?
The precipitation power of an electrolyte increases with ______.
Maximum coagulation power is in ____________.
Smoke is an example of ____________.
Which of the following is multimqlecular colloid?
Tyndall effect is useful ______.
Pumice stone is an example of ______.
Freshly prepared precipitate sometimes gets converted to colloidal solution by ______.
An emulsion cannot be broken by:
(i) heating
(ii) adding more amount of dispersion medium
(iii) freezing
(iv) adding emulsifying agent
Which of the following substances will precipitate the negatively charged emulsions?
(i) \[\ce{KCl}\]
(ii) glucose
(iii) urea
(iv) \[\ce{NaCl}\]
A colloid is formed by adding \[\ce{FeCl3}\] in excess of hot water. What will happen if excess sodium chloride is added to this colloid?
How do emulsifying agents stabilise the emulsion?
Colloids can
Toluene is oxidised to benzoic acid by
In which of the following, the Tyndall effect is not observed?
Blood may be purified by ______.
Starch is an example of which of the following type of colloid?
