Advertisements
Advertisements
प्रश्न
Distinguish between the three states of matter—solid, liquid, and gas on the basis of their molecular models
Advertisements
उत्तर
Solids:
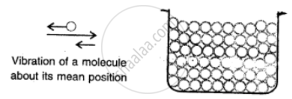
Here the molecules are very tighty packed that there is no or very less inteimolecular space and there is high intermolecular force of attraction (force of cohesion).
The molecules do not move about their mean position and thus solids have a definite shape and volume.
Liquids:
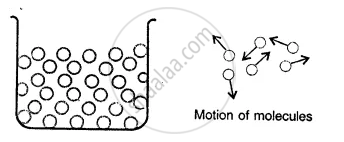
Here the molecules are less tightly packed as compared to solids and also there is lesser force of intermolecular attraction. The intermolecular distance is greater than that in the solids. Thus, they donot have a definite shape but acquire the shape of the vessel in which they are contained but have a definite volume at a given temperature.
Gases:
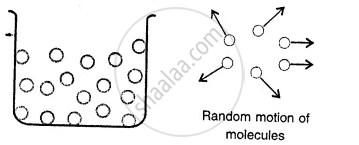
Here the molecules are far apart from each other i.e. have the greatest intermolecular distance which result into the weakest intermolecular forces of attraction. The molecules as are not bound by any strong force move about freely and thus gases do not have a definite shape and also do not have any definite volume.
APPEARS IN
संबंधित प्रश्न
Evaporation takes place over the entire mass of the liquid.
Evaporation takes place at ...............temperature.
Write true or false For the following Statement
The molecules in a substance arc in random motion.
Fill in the blank
A solid is ............ rigid.
How do the solids, liquids, and gases differ in their following properties.
(a) Size
(b) Shape
(c) Density
Give reason :
Liquids have a definite volume but no definite shape.
Write whether the following statement is true or false.
If the container in which a gas is collected has an opening, the gas will flow out and spread itself indefinitely.
Write true or false for the statement. Rewrite the false statement correctly.
Solids can be compressed easily.
Multiple choice question.
The process by which a solid changes into a liquid is called
Fill in the blanks:
Water is a matter because it has ______ and occupies ______.
