Advertisements
Advertisements
प्रश्न
Describe a method for the identification of primary, secondary and tertiary amines. Also write chemical equations of the reactions involved.
Advertisements
उत्तर
Benzenesulphonyl chloride (C6H5SO2Cl), which is also known as Hinsberg’s reagent, reacts with primary and secondary amines to form sulphonamides.
(i) The reaction of benzenesulphonyl chloride with primary amine yields N-ethylbenzenesulphonyl amide.
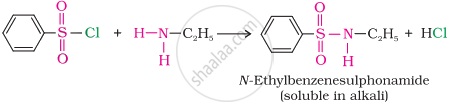
The hydrogen attached to nitrogen in sulphonamide is strongly acidic due to the presence of a strong electron-withdrawing sulphonyl group. Hence, it is soluble in alkali.
(ii) In the reaction with the secondary amine, N, N-diethylbenzenesulphonamide is formed.
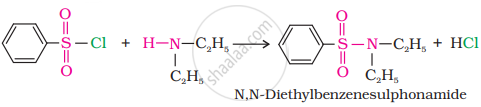
Since N, N-diethylbenzene sulphonamide does not contain any hydrogen atoms attached to nitrogen atoms, it is not acidic and hence insoluble in alkali.
(iii) Tertiary amines do not react with benzenesulphonyl chloride. This property of amines reacting with benzenesulphonyl chloride in a different manner is used for the distinction of primary, secondary and tertiary amines and also for the separation of a mixture of amines. However, these days benzenesulphonyl chloride is replaced by p-toluenesulphonyl chloride.
APPEARS IN
संबंधित प्रश्न
What is the action of nitrous acid on primary nitroalkane?
Distinguish between the following pairs of compounds: Aniline and N-methylaniline
What is the action of Benzene Sulphonyl Chloride on primary, secondary and tertiary
amines?
Describe Hinsberg method for the identification of primary, secondary and tertiary amines. Also write the chemical equations of the reactions involved.
Which of the following reactions will not give a primary amine?
On heating an aliphatic primary amine with chloroform and ethanolic potassium hydroxide, the organic compound formed is ______.
Identify the compound that will react with Hinsberg’s reagent to give a solid which dissolves in alkali.
Identify the compound that will react with Hinsberg’s reagent to give a solid which dissolves in alkali.
Compound A is converted to B on reaction with CHCl3 and KOH. The compound B is toxic and can be decomposed by C. A, B and C respectively are ______.
Isocyanide reaction involves the intermediate formation of ______.
The presence of primary amines can be confirmed by ______.
Give reasons:
(CH3)2NH is more basic than (CH3)3N in an aqueous solution.
Explain why (CH3)2NH is more basic than (CH3)3N in aqueous solution.
Carbylamine test is the distinguishing test for ______.
Given below are certain cations. Using inorganic qualitative analysis, arrange them in increasing group number from 0 to VI.
- Al3+
- Cu2+
- Ba2+
- Co2+
- Mg2+
Choose the correct answer from the options given below:
