Advertisements
Advertisements
प्रश्न
Define: Molecule
Advertisements
उत्तर
Molecule: A molecule can be defined as the smallest unit of an element or a compound which exhibits all the properties of that element or compound and has an independent existence. They are divisible into atoms.
APPEARS IN
संबंधित प्रश्न
Classify the following substances into compounds and mixtures:
Give one example for the following type of mixtures
solid-solid homogenous mixture.
Write true or false
All pure substances have characteristic melting and boiling points.
Correct the following statement:
CO and Co both represents cobalt.
Name: an Inert Gas
The important physical properties of substances are colour, odour, nature, density & solubility in water.
Name – two coloured gases (with their colours)
Write the names of elements from the following symbol:
Zn
Elements are broadly classified into metals and non-metals. State six general differences in the physical properties of metals and non-metals. State two metals and two non-metals which contradict with the general physical properties – giving reasons. State one difference in property between metalloids and noble gases.
The diagram represents fractional distillation for the separation of mixtures. Answer the following :
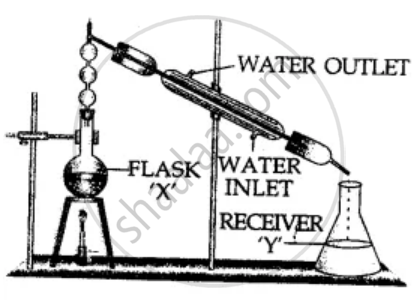
- Can two immiscible liquids be separated by this process?
- Separation of liquids by this process is based on which physical property?
- If methyl alcohol & water are to be separated, which liquid would remain in flask ‘X’ after condensation.
- Give a reason for the above answer.
- State the purpose of the fractionating column in the apparatus.
Select the correct answer from the choice given below to complete the sentence :
Dust in air is an example of _________ mixture.
