Advertisements
Advertisements
प्रश्न
Define the following term : Dehydrating agent
Advertisements
उत्तर
Dehydrating agent : The compound which has more affinity for water. It removes atoms of hydrogen and oxygen in the form of waterfrom the composition of a substance.
APPEARS IN
संबंधित प्रश्न
Give balanced chemical equations for the action of sulphuric acid on the following:
Sulphur
Which property of sulphuric acid is shown by the reaction of the concentrated sulphuric acid with carbon?
Why is H2SO4 is not used as a drying agent for H2S?
Name the Following:
The property used to prepare HCl and HNO3 from H2SO4.
Give reason for the following:
H2SO4 cannot be prepared by heating sodium sulphate with conc. HCl or HNO3
What would you see, when a crystal of CuSO4.5H2O is placed in concentrated sulphuric acid? Give reason to explain your observation.
Write the equation for the laboratory preparation of the following salts, using sulphuric acid.
Iron (II) sulphate from iron.
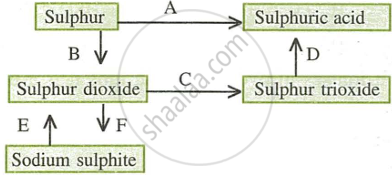
- Name the catalyst which helps in the conversion of sulphur dioxide to sulphur trioxide in step C.
- In the contact process for the manufacture of sulphuric acid, sulphur trioxide is not converted to sulphuric acid by reacting it with water. Instead a two-step procedure is used. Write the equations for the two steps involved in D.
- What type of substance will liberate sulphur dioxide from sodium sulphite in step E?
- Write the equation for the reaction by which sulphur dioxide is converted to sodium sulphite in step F.
A, B and C are the chemical properties of sulphuric acid:
A. Oxidizing agent
B. Dehydrating agent
C. Non-volatile acid
Match the following equations 1 to 3 to the above chemical properties of sulphuric acid.
1. \[\ce{KNO3 + H2SO4 -> KHSO4 + HNO3}\]
2. \[\ce{C12H22O11 -> 12C + 11H2O}\]
3. \[\ce{S + 2H2SO4 -> 2H2O + 3SO2}\]
Hydrogen chloride gas is prepared in the laboratory by the action of concentrated sulphuric acid on sodium chloride.
State the method of collection of the gas formed above.
