Advertisements
Advertisements
प्रश्न
Choose the correct option.
The homologous series of alcohols has general molecular formula ______.
पर्याय
CnH2n+1OH
Cn2n+2OH
CnH2n-2OH
CnH2nOH
Advertisements
उत्तर
The homologous series of alcohols has general molecular formula CnH2n+1OH.
APPEARS IN
संबंधित प्रश्न
Find out the most stable species from the following. Justify.
`bar"C""H"_3, bar"C""H"_2"Br", bar"C""Br"_3`
Identify the α-carbons in the following species and give the total number of α-hydrogen.
\[\ce{CH_3 - CH_2 - \overset{⊕}{C}H-CH_2 - CH_3}\]
Draw a resonance structure of the following:
Phenol
Draw a resonance structure of the following:
Benzaldehyde
Draw a resonance structure of the following:
Buta-1,3-diene
Distinguish between Inductive effect and resonance effect.
Write true or false. Correct the false statement.
Homolytic fission involves the unsymmetrical breaking of a covalent bond.
Write true or false. Correct the false statement.
Heterolytic fission results in the formation of free radicals.
Write true or false. Correct the false statement.
Aniline is a heterocyclic compound.
Predict the order of reactivity of the following compounds by SNl reaction mechanism.
\[\ce{\underset{\text{(I)}}{C6H5CH(C6H5)Cl}}\]
\[\ce{\underset{\text{(II)}}{C6H5CH2Cl}}\]
\[\ce{\underset{\text{(III)}}{C6H5C(CH3)(C6H5)Cl}}\]
Which of the following is NOT an electrophile?
Which of the following is the strongest nucleophile?
Which of the following alkyl groups shows maximum positive inductive effect?
The most unstable free radical among the following is:
The overlap of σ-p orbitals is called ____________.
Resonance is NOT exhibited by ____________.
How many pi bonds and sigma bonds are present in following molecule?
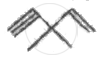
The most probable product in the reaction given below is:

Which element among the following does form pπ - pπ multiple bonds?
Arrange the following free radicals in order of decreasing stability.
- Methyl
- Vinyl
- Allyl
- Benzyl
Identify the α-carbons in the following species and give the total number of α-hydrogen.
\[\ce{CH3 - CH2 - \overset{⊕}{C}H - CH2 - CH3}\]
Identify the α - carbons in the following species and give the total number of α-hydrogen.
\[\ce{CH3 - CH2 - \overset{⊕}{C}H - CH2 - CH3}\]
Identify the α-carbons in the following species and give the total number of α-hydrogen.
\[\ce{CH2 = CH - CH2 - CH3}\]
Identify the α-carbons in the following species and give the total number of α-hydrogen in it.
CH2 = CH − CH2 − CH3
Identify the α-carbon in the following species and give the total number of α-hydrogen.
\[\ce{CH2 = CH - CH2 - CH3}\]
Identify the α-carbons in the following species and give the total number of α-hydrogen.
CH2 = CH - CH2 - CH3
Identify the α - carbons in the following species and give the total number of α-hydrogen in each.
\[\ce{CH3 - CH2 - \overset{\oplus}{C}H - CH2 - CH3}\]
Identify the α - carbon in the following species and give the total number of α-hydrogen.
\[\ce{CH2 = CH - CH2 - CH3}\]
