Advertisements
Advertisements
प्रश्न
With reference to the variation of properties in the Periodic Table, which of the following is generally true?
Atomic size increases from left to right across a period.
पर्याय
True
False
Advertisements
उत्तर
False
APPEARS IN
संबंधित प्रश्न
Why is the size of neon greater than fluorine?
Give reason for the following:
Argon atom is bigger than chlorine atom.
Write the name and symbol of the element from the description.
The atom having the smallest size.
Write the name and symbol of the element from the description.
The atom having the smallest atomic mass.
Fill in the blanks.
Size of the atoms ______ from left to right across a period and ______ on descending in a group of normal elements.
Which of the following is the correct order of atomic size?
Which of the following is the correct order of size?
Which among the following elements has the largest atomic radii?
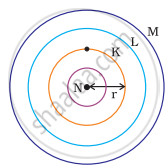
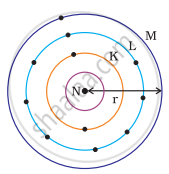
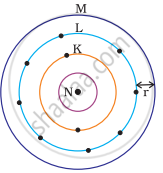
Which one of the following depict the correct representation of atomic radius(r) of an atom?
|
(i) |
(ii) |
|
(iii) |
(iv) |
This question refers to the elements of the Periodic Table with atomic numbers from 3 to 18. Some of the elements are shown by letters, but the letters are not the usual symbols of the elements.
| 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 |
| A | B | C | D | E | F | G | H |
| 11 | 12 | 13 | 14 | 15 | 16 | 17 | 18 |
| I | J | K | L | M | N | O | P |
Which of these have least atomic size in period 3?