Therefore, Benzaldehyde does NOT give Fehling’s test.
Advertisements
Advertisements
प्रश्न
Benzaldehyde does NOT show positive test with ______.
पर्याय
Schiff reagent
Tollens' reagent
Sodium bisulphite solution
Fehling solution
Advertisements
उत्तर
Benzaldehyde does NOT show positive test with Fehling solution.
Explanation:
Benzaldehyde is an aromatic aldehyde. Aromatic aldehydes do not reduce Fehling solution, so the test is negative. But they do give:
- Schiff test
- Tollens’ test
- Sodium bisulphite addition
APPEARS IN
संबंधित प्रश्न
Answer in brief.
What is the action of hydrazine on cyclopentanone in presence of KOH in ethylene glycol?
Write a note on Stephen reaction.
Explain haloform reaction with suitable example.
Write a reaction to distinguish acetaldehyde from acetone.
Which of the following is Schiff's reagent?
Which of the following compounds does NOT undergo aldol condensation?
\[\begin{array}{cc} \phantom{...}\ce{R}\phantom{.................}\ce{R}\phantom{..}\ce{H}\phantom{................}\\ \phantom{...}\backslash\phantom{..................}\backslash/\phantom{..........}\phantom{.......}\\ \ce{C = O ->[H2NNH2][KOH/glycol] C + N2 + H2O}\\ \phantom{}/\phantom{............}\phantom{.......}/\phantom{}\backslash\phantom{...............}\\ \phantom{}\ce{R}\phantom{..................}\ce{R}\phantom{..}\ce{H}\phantom{...............} \end{array}\]
Reaction is called ____________.
Compounds of general formula, are called ____________.
\[\begin{array}{cc} \phantom{...}\ce{R}\phantom{....}\ce{OR'}\phantom{}\\ \phantom{}\backslash\phantom{..}/\\ \ce{C}\\ \phantom{}/\phantom{..}\backslash\\ \phantom{...}\ce{H}\phantom{....}\ce{OR'}\phantom{} \end{array}\]
Which of the following is a Wolf - Kishner reduction?
Identify the product Y in the following series of reactions.
\[\ce{4-Nitrotoluene ->[(CH3CO)2O/CrO3][273 - 278 K] X ->[H3O^+][\Delta] Y}\]
Assertion: 2, 2-dimethyl propanoic acid does not give HVZ reaction.
Reason: 2-2, dimethyl propanoic acid does not have α-hydrogen atom.
\[\ce{Benzoic acid ->[i) NH3][ii) \Delta] A ->[NaOBr] B ->[NaNO2/HCl] C}\] ‘C’ is:
Which one of the following reaction is an example of disproportionation reaction.
Phenyl methanal is reacted with concentrated NaOH to give two products X and Y. X reacts with metallic sodium to liberate hydrogen X and Y are ____________.
Carboxylic acids have higher boiling points than aldehydes, ketones and even alcohols of comparable molecular mass. It is due to their ____________.
Identify A, B, C and D.
\[\ce{ethanoic acid ->[SOCl2] A ->[Pd/BaSO4] B ->[NaOH] C ->[][\Delta] D}\]
How will you convert benzaldehyde into the following compound?
Benzoic acid
What is the action of HCN on ethanal?
How will you prepare lactic acid from ethanol?
A substance C4H10O yields on oxidation a compound, C4H8O which gives an oxime and a positive iodoform test. The original substance on treatment with a conc. H2SO4 gives C4H8. The structure of the compound is ______.
Which of the following is most reactive towards the addition reaction of hydrogen cyanide to form the corresponding cyanohydrin?
What is the number of different products formed when mixture of ethanal and propanal reacts with aq. NaOH after warming?
Aldehydes are readily oxidised to yield carboxylic acids but ketones are inert to oxidation. Which is the most likely explanation regarding this difference in reactivity?
Write the structure of the major product of the aldol condensation of benzaldehyde with acetone.
Write the structure of the major product of the aldol condensation of benzaldehyde with acetone.
How will you prepare acetic anhydride from acetic acid.
How will you prepare Acetic anhydride from acetic acid ?
What is IUPAC name for
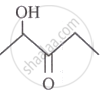 ?
?
