Advertisements
Advertisements
प्रश्न
Balance the following redox equation by half-reaction method.
\[\ce{Bi(OH)_{3(s)} + SnO^2-_{2(aq)}->SnO^2-_{3(aq)} + Bi^_{(s)}(basic)}\]
Advertisements
उत्तर
\[\ce{Bi(OH)_{3(s)} + SnO^2-_{2(aq)}->SnO^2-_{3(aq)} + Bi^_{(s)}}\]
Step 1: Write the unbalanced equation for the redox reaction. Assign the oxidation number to all the atoms in reactants and products.
Divide the equation into two half equations.
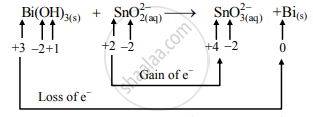
- Oxidation half-reaction: \[\ce{SnO^2-_{2(aq)}->SnO^2-_{3(aq)}}\]
- Reduction half-reaction: \[\ce{Bi(OH)_{3(s)}->Bi_{(s)}}\]
Step 2: Balance half equations for O atoms by adding H2O to the side with fewer O atoms. Add 1 H2O to the left side of the oxidation half equation and 3H2O to the right side of the reduction half equation.
- Oxidation: \[\ce{SnO^2-_{2(aq)} + H2O_{(l)}->SnO^2_{3(aq)}}\]
- Reduction: \[\ce{Bi(OH)_{3(s)}->Bi_{(s)} + 3H2O_{(l)}}\]
Step 3: Balance H+ atoms by adding H+ ions to the side with less H. Hence, add 2H+ ions to the right side of the oxidation half equation and 3H+ ions to the left side of the reduction half equation.
- Oxidation: \[\ce{SnO^2-_{2(aq)} + H2O_{(l)}->SnO^2-_{3(aq)} + 2H^+_{( aq)}}\]
- Reduction: \[\ce{Bi(OH)_{3(s)} + 3H^+_{( aq)}->Bi_{(s)} + 3H2O_{(l)}}\]
Step 4: Now add 2 electrons to the right side of the oxidation half equation and 3 electrons to the left side of the reduction half equation to balance the charges.
- Oxidation: \[\ce{SnO^2-_{2(aq)} + H2O_{(l)}->SnO^2-_{3(aq)} + 2H^+_{( aq)} + 2e-}\]
- Reduction: \[\ce{Bi(OH)_{3(s)} + 3H^+_{( aq)} + 3e- ->Bi_{(s)} + 3H2O_{(l)}}\]
Step 5: Multiply the oxidation half equation by 3 reduction half equation by 2 to equalize the number of electrons in two half equations.
Then add two half equations.
- Oxidation: \[\ce{3SnO^2-_{2(aq)} + 3H2O_{(l)}->3SnO^2-_{3(aq)} + 6H^+_{( aq)} + 6e-}\]
- Reduction: \[\ce{2Bi(OH)_{3(s)} + 6H^+_{( aq)} + 6e- ->2Bi_{(s)} + 6H2O_{(l)}}\]
Add two half equations:
\[\ce{2Bi(OH)_{3(s)} + 3SnO^2-_{2(aq)}->3SnO^2-_{3(aq)} + 2Bi_{(s)} + 3H2O_{(l)}}\]
A reaction occurs in a basic medium. However, H+ ions cancel out, and the reaction is balanced. Hence, no need to add OH− ions. The equation is balanced in terms of number of atoms and the charges
Hence, balanced equation: \[\ce{2Bi(OH)_{3(s)} + 3SnO^2-_{2(aq)}->3SnO^2-_{3(aq)} + 2Bi_{(s)} + 3H2O_{(l)}}\]
APPEARS IN
संबंधित प्रश्न
Consider the reaction:
\[\ce{O3(g) + H2O2(l) → H2O(l) + 2O2(g)}\]
Why it is more appropriate to write these reaction as:
\[\ce{O3(g) + H2O2 (l) → H2O(l) + O2(g) + O2(g)}\]
Also, suggest a technique to investigate the path of the redox reactions.
Whenever a reaction between an oxidising agent and a reducing agent is carried out, a compound of lower oxidation state is formed if the reducing agent is in excess and a compound of higher oxidation state is formed if the oxidising agent is in excess. Justify this statement giving three illustrations.
Balance the following equation in the basic medium by ion-electron method and oxidation number methods and identify the oxidising agent and the reducing agent.
\[\ce{N2H4(l) + ClO^-_3 (aq) → NO(g) + Cl–(g)}\]
Balance the following equation in basic medium by ion-electron method and oxidation number methods and identify the oxidising agent and the reducing agent.
\[\ce{Cl_2O_{7(g)} + H_2O_{2(aq)} -> ClO-_{2(aq)} + O_{2(g)} + H+_{(aq)}}\]
The Mn3+ ion is unstable in solution and undergoes disproportionation to give Mn2+, MnO2, and H+ ion. Write a balanced ionic equation for the reaction.
Chlorine is used to purify drinking water. Excess of chlorine is harmful. The excess of chlorine is removed by treating with sulphur dioxide. Present a balanced equation for this redox change taking place in water.
Choose the correct option.
For the following redox reactions, find the correct statement.
\[\ce{Sn^{2⊕} + 2Fe^{3⊕}->Sn^{4⊕} + 2Fe^{2⊕}}\]
Balance the following reaction by oxidation number method.
\[\ce{Cr2O^2-_{7(aq)} + SO^2-_{3(aq)}->Cr^3+_{ (aq)} + SO^2-_{4(aq)}(acidic)}\]
Balance the following reaction by oxidation number method.
\[\ce{H2SO4_{(aq)} + C_{(s)} -> CO2_{(g)} + SO2_{(g)} + H2O_{(l)}(acidic)}\]
Balance the following reaction by oxidation number method.
\[\ce{Bi(OH)_{3(s)} + Sn(OH)^-_{3(aq)}->Bi_{(s)} + Sn(OH)^2-_{6(aq)}(basic)}\]
Balance the following redox equation by half-reaction method.
\[\ce{H2C2O_{4(aq)} + MnO^-_{4(aq)}->CO2_{(g)} + Mn^2+_{( aq)}(acidic)}\]
Identify coefficients 'x' and 'y' for the following reaction.
\[\ce{{x}H2O2_{(aq)} + ClO^-_{4(aq)} -> 2O2_{(g)} + ClO^-_{2(aq)} + {y}H2O_{(l)}}\]
What is the change in oxidation number of Sulphur in following reaction?
\[\ce{MnO^-_{4(aq)} + SO^{2-}_{3(aq)} -> MnO^{2-}_{4(aq)} + SO^{2-}_{4(aq)}}\]
Identify the oxidising agent in the following reaction:
\[\ce{CH4_{(g)} + 2O2_{(g)} -> CO2_{(g)} + 2H2O_{(l)}}\]
Write balanced chemical equation for the following reactions:
Permanganate ion \[\ce{(MnO^{-}4)}\] reacts with sulphur dioxide gas in acidic medium to produce \[\ce{Mn^{2+}}\] and hydrogen sulphate ion.
Write balanced chemical equation for the following reactions:
Reaction of liquid hydrazine \[\ce{(N2H4)}\] with chlorate ion \[\ce{(ClO^{-}3)}\] in basic medium produces nitric oxide gas and chloride ion in gaseous state.
Write balanced chemical equation for the following reactions:
Dichlorine heptaoxide \[\ce{(Cl2O7)}\] in gaseous state combines with an aqueous solution of hydrogen peroxide in acidic medium to give chlorite ion \[\ce{(ClO^{-}2)}\] and oxygen gas. (Balance by ion-electron method)
Balance the following equations by the oxidation number method.
\[\ce{I2 + NO^{-}3 -> NO2 + IO^{-}3}\]
Identify the redox reactions out of the following reactions and identify the oxidising and reducing agents in them.
\[\ce{4NH3 (g) + 3O2 (g) -> 2N2 (g) + 6H2O (g)}\]
Balance the following ionic equations.
\[\ce{Cr2O^{2-}7 + H^{+} + I- -> Cr^{3+} + I2 + H2O}\]
Balance the following ionic equations.
\[\ce{MnO^{-}4 + H^{+} + Br^{-} -> Mn^{2+} + Br2 + H2O}\]
In acidic medium, reaction, \[\ce{MNO^-_4 → Mn^2+}\] an example of ____________.
In the reaction of oxalate with permanganate in an acidic medium, the number of electrons involved in producing one molecule of CO2 is ______.
Consider the following reaction:
\[\ce{xMnO^-_4 + yC2O^{2-}_4 + zH^+ -> xMn^{2+} + 2{y}CO2 + z/2H2O}\]
The values of x, y, and z in the reaction are, respectively:
\[\ce{H2O2 -> 2H^+ + O2 + 2e^-}\]; E0 = −0.68 V.
This equation represents which of the following behaviour of H2O2?
