Advertisements
Advertisements
प्रश्न
Account for the following:
Aniline does not undergo Friedel-Crafts reaction.
Advertisements
उत्तर
Aniline being a Lewis base reacts with Lewis acid AlCl3 to form a salt.
\[\ce{C6H5\overset{\bullet\bullet}{N}H2 + AlCl3 -> C6H5\overset{+}{N}H2AlCl^-_3}\]
Due to the presence of a positive charge on N-atom in the salt, the group \[\ce{-\overset{+}{N}H2AlCl^-_3}\] acts as a strongly deactivating group. As a result, it reduces the electron density in the benzene ring and inhibits the electrophilic substitution reaction. Therefore, aniline does not undergo the Friedel-Crafts reaction.
संबंधित प्रश्न
The method by which aniline cannot be prepared is ____________.
\[\ce{CH3CH2Br ->[aq NaOH][\Delta] A ->[KMnO4/H^+][\Delta] B ->[NH3][\Delta] C ->[Br2/NaOH] D}\] ‘D’ is:
Which of the following reaction is not correct.
Secondary nitro alkanes react with nitrous acid to form ____________.
Among the following, the reaction that proceeds through an electrophilic substitution is:
Identify A, B and C.
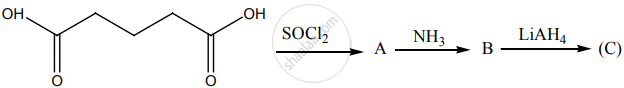
Account for the following
Aniline does not undergo Friedel–Crafts reaction
Account for the following.
Aniline does not undergo Friedel–Crafts reaction
Account for the following:
Aniline does not undergo Friedel - Crafts reaction.
Account for the following:
Aniline does not undergo Friedel – Crafts reaction.
