Advertisements
Advertisements
प्रश्न
A student heats a beaker containing ice and water. He measures the temperature of the content of the beaker as a function of time. Which of the following (Fig. 1.1) would correctly represent the result? Justify your choice.
पर्याय
Advertisements
उत्तर
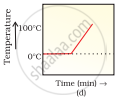
Since ice and water are in equilibrium, the temperature would be zero. When we heat the mixture, ‘energy supplied is utilised in melting the ice and the temperature does not change till the ice melts because of latent heat of fusion. On further heating the temperature of the water would increase. Therefore (d) is the correct option.
APPEARS IN
संबंधित प्रश्न
Explain the term Melting Point.
Describe an experiment to demonstrate that a substance absorbs heat during melting without change in its temperature.
State the melting point of ice.
The fixed temperature at which matter changes state from solid to liquid is called ______.
Analogy:
Melting: ice into water on heating,
freezing: _______.
Conversion of solid state to liquid state is called fusion; what is meant by latent heat of fusion?
It is a hot summer day, Priyanshi and Ali are wearing cotton and nylon clothes respectively. Who do you think would be more comfortable and why?
In an endothermic process, the speed of the molecules is ______ hence they move faster.
A puddle of water gets pooled around the glass of ice cream or a glass of ice cubes. When it is kept at room temperature. Give reason.
Define the following:
Melting or fusion: