Please select a subject first
Advertisements
Advertisements
Calculate spin only magnetic moment of divalent cation of transition metal with atomic number 25.
Concept: undefined >> undefined
Neutral solutions have the pH of ______.
Concept: undefined >> undefined
Advertisements
Presence of disulphide link gives rise to which structure of protein?
Concept: undefined >> undefined
Aluminium crystallises in a cubic close-packed structure with a unit cell edge length of 353.6 pm. What is the radius of Al atom? How many unit cells are there in 1.00 cm3 of Al?
Concept: undefined >> undefined
Convert the following.
p-Nitrochlorobenzene to p-nitrophenol
Concept: undefined >> undefined
The pH of a weak monoacidic base is 11.2, and its OH– ion concentration is ______.
Concept: undefined >> undefined
The spin only magnetic moment of Mn2+ ion is ______.
Concept: undefined >> undefined
The product of following reaction is
\[\ce{CH3 - CH = CH - CH2 - CHO ->[i) LiAlH4][ii) H3O+]}\] ______?
Concept: undefined >> undefined
Explain the dehydrohalogenation reaction of 2-chlorobutane.
Concept: undefined >> undefined
Give the classification of alloys with examples.
Concept: undefined >> undefined
Complete the following reaction giving major products.
\[\begin{array}{cc}
\ce{CH3}\phantom{.......}\\
|\phantom{.........}\\
\ce{CH3 - c - CH2 - Cl ->[Na/dry ether]}\\
|\phantom{.........}\\
\ce{CH3}\phantom{.......}
\end{array}\]
Concept: undefined >> undefined
Complete the following reactions giving a major product.
\[\begin{array}{cc}
\ce{CH3}\phantom{................}\\
|\phantom{..................}\\
\ce{CH3 - C - CH2 - Cl ->[Na/dry ether] A}\\
|\phantom{..................}\\
\ce{CH3}\phantom{................}
\end{array}\]
Concept: undefined >> undefined
Complete the following reaction giving a major product.
\[\begin{array}{cc}
\ce{CH3}\phantom{.................}\\
|\phantom{...................}\\
\ce{CH3 - C - CH2 - Cl->[Na/dry ether]A}\\
|\phantom{..................}\\
\ce{CH3}\phantom{................}
\end{array}\]
Concept: undefined >> undefined
What two uses of the alloys.
Concept: undefined >> undefined
Why dissociation of HCN is suppressed by the addition of HCL?
Concept: undefined >> undefined
What is zeroth order reaction? Derive its integrated rate Law. What are the units of rate constant?
Concept: undefined >> undefined
Give two reactions showing oxidising property of concentrated H2SO4. Write the structures of HClO3 and HClO4.
Concept: undefined >> undefined
A gas is allowed to expand in a well insulated container against a constant external pressure of 2.5 Bar from an initial volume of 2.5L to a final volume of 4.5L The change in internal energy, ΔU of the gas will be ______.
Concept: undefined >> undefined
The coordination number of cobalt in the complex [Co(en)2Br2]Cl2 is ______.
Concept: undefined >> undefined
Observe the following and answer the questions given below:
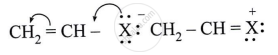
Name the type of halogen derivative.
Concept: undefined >> undefined
