Please select a subject first
Advertisements
Advertisements
Provide the stock notation for the following compound:
FeO
Concept: undefined >> undefined
Provide the stock notation for the following compound:
Fe2O3
Concept: undefined >> undefined
Advertisements
Provide the stock notation for the following compound:
CuO
Concept: undefined >> undefined
Provide the stock notation for the following compound:
MnO
Concept: undefined >> undefined
Assign oxidation number atom in the following species.
\[\ce{Cr(OH)^Θ_4}\]
Concept: undefined >> undefined
Assign oxidation number atom in the following species.
Na2S2O3
Concept: undefined >> undefined
Assign oxidation number atom in the following species.
H3BO3
Concept: undefined >> undefined
Which of the following redox couple is a stronger oxidizing agent?
Cl2 (E0 = 1.36 V) and Br2 (E0 = 1.09 V)
Concept: undefined >> undefined
Which of the following redox couple is a stronger oxidizing agent?
\[\ce{MnO^Θ_4}\](E0 = 1.51 V) and \[\ce{Cr2O^{2Θ}_7}\](E0 = 1.33 V)
Concept: undefined >> undefined
Which of the following redox couple is a stronger reducing agent?
Li (E0 = - 3.05 V) and Mg (E0 = - 2.36 V)
Concept: undefined >> undefined
Which of the following redox couple is a stronger reducing agent?
Zn (E0 = - 0.76 V) and Fe (E0 = - 0.44 V)
Concept: undefined >> undefined
Write condensed formulae and bond line formulae for the following structure.
\[\begin{array}{cc}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{...}\\\phantom{...}|\phantom{....}|\phantom{....}|\phantom{....}|\phantom{...}\\\ce{H - C - C - C - C - H}\\|\phantom{....}|\phantom{....}|\phantom{....}|\\
\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\\\end{array}\]
Concept: undefined >> undefined
Write condensed formulae and bond line formulae for the following structure.
\[\begin{array}{cc}\ce{H}\phantom{...}\ce{H}\\|\phantom{....}|\\\ce{N ≡ C - C - C - C ≡ N}\\
|\phantom{....}|\\\ce{H}\phantom{...}\ce{H}\end{array}\]
Concept: undefined >> undefined
Write condensed formulae and bond line formulae for the following structure.
\[\begin{array}{cc}
\phantom{.......}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{.....}\ce{O}\\
\phantom{.......}|\phantom{....}|\phantom{....}| \phantom{....}//\\
\ce{H - C - C - C - C}\\
\phantom{......}|\phantom{....}|\phantom{....}| \phantom{.....}\backslash\\
\phantom{........}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{.....}\ce{OH}
\end{array}\]
Concept: undefined >> undefined
Write dash formula for the following bond line formula.

Concept: undefined >> undefined
Write dash formulae for the following bond line formulae.

Concept: undefined >> undefined
Write dash formulae for the following bond line formulae.
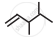
Concept: undefined >> undefined
Write dash formulae for the following bond line formulae.
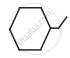
Concept: undefined >> undefined
Complete the following table:
Assign oxidation number to the underlined species and write Stock notation of compound
| Compound | Oxidation number | Stock notation |
| AuCl3 | ||
| SnCl2 | ||
| \[\ce{\underline{{V}}_2O^{4-}_{7}}\] | ||
| \[\ce{\underline{{Pt}}Cl^2-_6}\] | ||
| H3AsO3 |
Concept: undefined >> undefined
Write bond-line formulae and condensed formulae for the following compound.
3-methyloctane
Concept: undefined >> undefined
