Advertisements
Advertisements
प्रश्न
Justify that the following reaction is redox reaction; identify the species oxidized/reduced, which acts as an oxidant and which acts as a reductant.
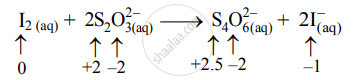
\[\ce{I2_{(aq)} + 2S2O^{2-}_{3(aq)}->S4O^{2-}_{6(aq)} + 2I^-_{ (aq)}}\]
Advertisements
उत्तर
\[\ce{I2_{(aq)} + 2S2O^{2-}_{3(aq)}->S4O^{2-}_{6(aq)} + 2I^-_{ (aq)}}\]
- Write oxidation number of all the atoms of reactants and products.
- Identify the species that undergoes a change in oxidation number.
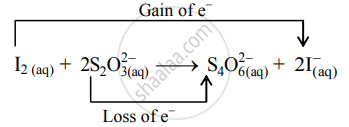
- The oxidation number of S increases from +2 to +2.5 and that of I decreases from 0 to –1. Because oxidation number of one species increases and that of the other decreases, the reaction is a redox reaction.
- The oxidation number of S increases by loss of electrons and therefore, S is a reducing agent and itself is oxidized. On the other hand, the oxidation number of I decreases by a gain of electrons, and therefore, I is an oxidizing agent and itself is reduced.
Result:
- The given reaction is a redox reaction.
- Oxidant/oxidizing agent (Reduced species): I2
- Reductant/reducing agent (Oxidized species): \[\ce{S2O^2-_3}\]
APPEARS IN
संबंधित प्रश्न
Choose the correct option.
Which of the following halogens does always show oxidation state -1?
In which chemical reaction does carbon exhibit variation of oxidation state from - 4 to + 4? Write a balanced chemical reaction.
Calculate the oxidation number of the underlined atom.
Cr2O72−
Calculate the oxidation number of the underlined atom.
NaH2PO4
Identify the following pair of species is in its oxidized state?
\[\ce{O2/O^2-}\]
Identify the following pair of species is in its oxidized state?
Cl2/Cl−
Provide the stock notation for the following compound:
Fe2O3
Which of the following redox couple is a stronger oxidizing agent?
\[\ce{MnO^Θ_4}\](E0 = 1.51 V) and \[\ce{Cr2O^{2Θ}_7}\](E0 = 1.33 V)
Oxidation state of Xe in XeOF4 is ____________.
What is the oxidation number of Mn in \[\ce{MnO^{2-}_4}\] ion?
In the reaction,
\[\ce{MnO^{-1}_4 (aq) + Br^{-1}(aq) -> MnO2(s) + BrO^{-1}_3(aq)}\]
the correct change in oxidation number of the species involved is ______.
Which of the following is NOT an example of redox reaction?
In the following reaction, the oxidation number of Cr changes.
\[\ce{ClO^-_{( aq)} + Cr(OH)^-_{4(aq)} -> CrO^{2-}_{4(aq)} + Cl^-_{( aq)} (basic)}\]
Identify the strongest oxidising agent.
\[\ce{Na^+ + e^- -> Na}\]; E0 = −2.714 V
\[\ce{Pt^{2+} + 2e^- -> Pt}\]; E0 = +1.200 V
\[\ce{I2 + 2e^- -> 2I^-}\]; E0 = + 0.535 V
\[\ce{Co^{2+} + 2e^- -> Co}\]; 0 = −0.280 V
The oxidation number of sulphur in S8 molecule is ______.
Stock notations are used to specify the oxidation numbers of ____________.
The sum of oxidation states of all atoms in \[\ce{SnO^{2-}_2}\] and CO2 are ____________ respectively.
The oxidation state of phosphorous in Mg2P2O7 is ______.
Which among the following pair of elements show highest oxidation state +7 in their different compounds?
The oxidation number of Cr in \[\ce{Cr(OH)^-_4}\] ion is ______.
The oxidation number of phosphorous in Ba(H2PO2)2 is ______.
What is the oxidation number of Cr in K2Cr2O7?
The brown ring complex is formulated as [Fe(H2O)5NO]SO4. The oxidation number of iron is ______.
The oxidation number of oxygen in oxygen difluoride (OF2) and dioxygen difluoride (O2F2) respectively is ______.
What is the average oxidation number of sulphur in tetrathionate ion?
Find CORRECT statement for following reaction.
\[\ce{2Ag+ + Cu -> 2Ag + Cu^2+}\]
Which of the following statements are INCORRECT?
- All the transition metals except scandium form MO oxides which are ionic.
- The highest oxidation number corresponding to the group number in transition metals oxides is attained in Sc2B5 to Mn2B7.
- Basic character increases from V2B5 to V2B4 to V2B5.
- V2B4 dissolves in acids to give VO43– salts.
- CrO is basic but Cr2B5 is atmospheric.
Choose the correct answer from the options given below:
