Advertisements
Advertisements
प्रश्न
Justify the following reaction as a redox reaction.
\[\ce{2Na_{(s)} + S_{(s)} -> Na2S_{(s)}}\]
Find out the oxidizing and reducing agents.
Advertisements
उत्तर
- A redox reaction can be described as electron transfer, as shown below:
\[\ce{2Na_{(s)} + S_{(s)}->2Na^+ + S^2-}\] - Charge development suggests that each sodium atom loses one electron to form Na+ and the sulphur atom gains two electrons to form S2–. This can be represented as follows:
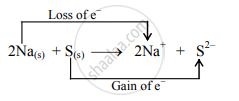
- When Na is oxidized to Na2S, the neutral Na atom loses electrons to form Na+ in Na2S while the elemental sulphur gains electrons and forms S2– in Na2S.
- Each of the above steps represents a half-reaction which involves electron transfer (loss or gain).
- The sum of these two half-reactions, or the overall reaction, is a redox reaction.
- An oxidizing agent is an electron acceptor, and hence, S is an oxidizing agent. A reducing agent is an electron donor, and hence, Na is a reducing agent.
APPEARS IN
संबंधित प्रश्न
Choose the correct option.
Oxidation numbers of Cl atoms marked as Cla and Clb in CaOCl2 (bleaching powder) are
\[\begin{array}{cc} \ce{Cl^{{a}}}\phantom{.} \\/\phantom{...} \\ \ce{Ca}\phantom{......} \\ \backslash\phantom{..} \\\phantom{........} \ce{O-Cl^{{b}}}\phantom{.} \end{array}\]
Choose the correct option.
The coefficients p, q, r, s in the reaction \[\ce{{p}Cr2O7^{2Θ} + {q}Fe^{2⊕}->{r}Cr^{3⊕} + {s}Fe^{3⊕} + H2O}\] respectively are:
Choose the correct option.
Oxidation number of carbon in H2CO3 is
Choose the correct option.
Which is the correct stock notation for manganese dioxide?
Calculate the oxidation number of the underlined atom.
HNO3
Calculate the oxidation number of the underlined atom.
H2S4O6
Calculate the oxidation number of the underlined atom.
Cr2O72−
Identify the following pair of species is in its oxidized state?
\[\ce{O2/O^2-}\]
Provide the stock notation for the following compound:
HAuCl4
Assign oxidation number atom in the following species.
\[\ce{Cr(OH)^Θ_4}\]
Oxidation state of Xe in XeOF4 is ____________.
What is the oxidation number of Mn in \[\ce{MnO^{2-}_4}\] ion?
In the following reaction, the oxidation number of Cr changes.
\[\ce{ClO^-_{( aq)} + Cr(OH)^-_{4(aq)} -> CrO^{2-}_{4(aq)} + Cl^-_{( aq)} (basic)}\]
Identify the strongest oxidising agent.
\[\ce{Na^+ + e^- -> Na}\]; E0 = −2.714 V
\[\ce{Pt^{2+} + 2e^- -> Pt}\]; E0 = +1.200 V
\[\ce{I2 + 2e^- -> 2I^-}\]; E0 = + 0.535 V
\[\ce{Co^{2+} + 2e^- -> Co}\]; 0 = −0.280 V
The oxidation number of sulphur in S8 molecule is ______.
Match the following.
| Compound | Oxidation no. of underlined element |
| i. \[\ce{\underline{C}_4H4O^{2-}_6}\] | a. +2.5 |
| ii. \[\ce{\underline{N}_3H}\] | b. +1.5 |
| iii. \[\ce{Mg2\underline{P}_2O7}\] | c. +5 |
| iv. \[\ce{Na2\underline{S}_4O6}\] | d. `-1//3` |
In which of the following, oxidation number of oxygen is +2?
Oxidation state of nitrogen in nitric oxide is ______.
The sum of oxidation states of all atoms in \[\ce{SnO^{2-}_2}\] and CO2 are ____________ respectively.
Which of the following is INCORRECT?
What is the oxidation number of Cr in K2Cr2O7?
In which of the compounds does ‘manganese’ exhibit the highest oxidation number?
