Advertisements
Advertisements
प्रश्न
Justify the following reaction as a redox reaction.
\[\ce{2Na_{(s)} + S_{(s)} -> Na2S_{(s)}}\]
Find out the oxidizing and reducing agents.
Advertisements
उत्तर
- A redox reaction can be described as electron transfer, as shown below:
\[\ce{2Na_{(s)} + S_{(s)}->2Na^+ + S^2-}\] - Charge development suggests that each sodium atom loses one electron to form Na+ and the sulphur atom gains two electrons to form S2–. This can be represented as follows:
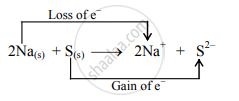
- When Na is oxidized to Na2S, the neutral Na atom loses electrons to form Na+ in Na2S while the elemental sulphur gains electrons and forms S2– in Na2S.
- Each of the above steps represents a half-reaction which involves electron transfer (loss or gain).
- The sum of these two half-reactions, or the overall reaction, is a redox reaction.
- An oxidizing agent is an electron acceptor, and hence, S is an oxidizing agent. A reducing agent is an electron donor, and hence, Na is a reducing agent.
APPEARS IN
संबंधित प्रश्न
Choose the correct option.
Oxidation number of oxygen in superoxide is
Choose the correct option.
The process \[\ce{SO2->S2Cl2}\] is
In which reaction does nitrogen exhibit variation of oxidation state from –3 to +5?
Calculate the oxidation number of the underlined atom.
H2SO4
Calculate the oxidation number of the underlined atom.
H3PO3
Identify the following pair of species is in its oxidized state.
Mg/Mg2+
Identify the following pair of species is in its oxidized state?
\[\ce{O2/O^2-}\]
Provide the stock notation for the following compound:
Tl2O
Provide the stock notation for the following compound:
FeO
Provide the stock notation for the following compound:
CuO
Assign oxidation number atom in the following species.
Na2S2O3
Which of the following redox couple is a stronger oxidizing agent?
Cl2 (E0 = 1.36 V) and Br2 (E0 = 1.09 V)
Which of the following redox couple is a stronger reducing agent?
Zn (E0 = - 0.76 V) and Fe (E0 = - 0.44 V)
In the following reaction, the oxidation number of Cr changes.
\[\ce{ClO^-_{( aq)} + Cr(OH)^-_{4(aq)} -> CrO^{2-}_{4(aq)} + Cl^-_{( aq)} (basic)}\]
Stock notations are used to specify the oxidation numbers of ____________.
The sum of oxidation states of all atoms in \[\ce{SnO^{2-}_2}\] and CO2 are ____________ respectively.
When methane undergoes combustion in air, the oxidation number of C ____________.
The sum of oxidation states of all atoms in \[\ce{Cr2O^{2-}_7}\] ion is ______.
What is the oxidation number of Carbon in K2C2O4?
The oxidation state of phosphorous in Mg2P2O7 is ______.
The oxidation number of Cr in \[\ce{Cr(OH)^-_4}\] ion is ______.
The oxidation number of phosphorous in Ba(H2PO2)2 is ______.
Which of the following explanation is correct about the given below reaction?
\[\ce{Cr2O^2-_7 + H2O -> 2CrO^2-_4 + 2H+}\]
The oxidation number of oxygen in oxygen difluoride (OF2) and dioxygen difluoride (O2F2) respectively is ______.
Which of the following changes exhibit that nitrogen undergoes oxidation?
Find CORRECT statement for following reaction.
\[\ce{2Ag+ + Cu -> 2Ag + Cu^2+}\]
