Advertisements
Advertisements
Explain the application of heating effect of electric current in an electric bulb with a diagram.
Concept: undefined >> undefined
If each two concave lenses of focal length 30 cm are kept in contact with each other what will be the power of combination?
Concept: undefined >> undefined
Advertisements
An object is placed at a distance of 10 cm from a convex lens of focal length 12 cm, find out at what distance image will be formed from the lens and what is the nature of image.
Concept: undefined >> undefined
5 cm high object is placed at a distance of 10 cm from a converging lens of focal length of 20 cm. Determine the position, size and type of the image.
Concept: undefined >> undefined
Two convex lenses of focal length 30 cm and 10 cm each are kept in contact with each other. Find the power of their combination.
Concept: undefined >> undefined
Explain the hydraulic separation method with a neat labelled diagram.
Concept: undefined >> undefined
Identify the following method of concentration of ores and explain briefly.
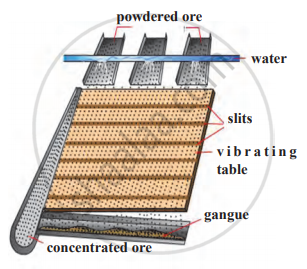
Concept: undefined >> undefined
Explain the froth floatation method with a neat labelled diagram.
Concept: undefined >> undefined
Complete the following flowchart.

Concept: undefined >> undefined
Complete the following table for the homologous series of alkanes.
| Name | Molecular formula | Condensed structural formula | Number of carbon atom | Number of -CH2- units | Boiling point °C |
| Methane | CH4 | CH4 | 1 | 1 | -162 |
| Ethane | C2H6 | CH3–CH3 | 2 | 2 | -88.5 |
| Propane | C3H8 | CH3–CH2–CH3 | 3 | 3 | -42 |
| Butane | C4H10 | CH3–CH2–CH2–CH3 | ______ | ______ | 0 |
| Pentane | C5H12 | CH3–CH2–CH2–CH2–CH3 | ______ | ______ | 36 |
| Hexane | C6H14 | CH3–CH2–CH2–CH2–CH2–CH3 | ______ | ______ | 69 |
Concept: undefined >> undefined
Complete the following table for homologous series of alcohols.
| Name | Molecular formula | Condensed structural formula | Number of carbon atom | Number of -CH2- units | Boiling point °C |
| Methanol | CH4O | CH3-OH | 1 | 1 | 63 |
| Ethanol | C2H6O | CH3–CH2-OH | 2 | 2 | 78 |
| Propanol | C3H8O | CH3–CH2–CH2-OH | ______ | ______ | 97 |
| Butanol | C4H10O | CH3–CH2–CH2–CH2–OH | ______ | ______ | 118 |
Concept: undefined >> undefined
Complete the following table for homologous series of Alkenes.
| Name | Molecular formula | Condensed structural formula | Number of carbon atom | Number of -CH2- units | Boiling point °C |
| Ethene | C2H4 | CH2 = CH2 | 2 | 0 | -102 |
| Propene | C3H6 | CH3–CH = CH2 | 3 | 1 | -48 |
| 1-Butene | C4H8 | CH3–CH2–CH = CH2 | ______ | ______ | -6.5 |
| 1-Pentene | C5H10 | CH3–CH2–CH2–CH = CH2 | ______ | ______ | 30 |
Concept: undefined >> undefined
Observe the structural formula and answer the following questions.
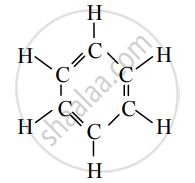
- Write the name of the given hydrocarbon.
- The given hydrocarbon is included in which type of hydrocarbon?
- What is the kind of compounds with the above characteristic structure called?
Concept: undefined >> undefined
Complete the following chart by using examples given in brackets.
(isobutylene, cyclohexane, propane, cyclohexene, cyclopentane, benzene, propyne, isobutane, propene)
| Straight chain hydrocarbons | Branched chain hydrocarbons | Cyclic hydrocarbons |
Concept: undefined >> undefined
Observe the figure and write the answers to the questions asked.

- The construction shown in the figure is of which device?
- Explain the principle on which this device works.
- Write any three uses of this device.
Concept: undefined >> undefined
Observe the figure and name and explain in brief the following method:

Concept: undefined >> undefined
Observe the figure and name and explain in brief the following method:

Concept: undefined >> undefined
Identify the type of chemical reaction given below:
\[\ce{CuSO4 + Fe -> FeSO4 + Cu}\]
Concept: undefined >> undefined
Explain the following term:
Metallurgy
Concept: undefined >> undefined
Define Homologous series.
Concept: undefined >> undefined
