Advertisements
Advertisements
Name the following compound according to IUPAC system.
\[\begin{array}{cc}
\ce{CH3 - C = C - CH2OH}\\
\phantom{}|\phantom{....}|\phantom{....}\\
\phantom{}\ce{CH3}\phantom{.}\ce{Br}\phantom{...}
\end{array}\]
Concept: undefined >> undefined
Write IUPAC name of the following compound:
\[\begin{array}{cc}
\phantom{................}\ce{CH3}\\
\phantom{.............}|\\
\ce{CH3 - CH - CH - C - CH3}\\
|\phantom{......}|\phantom{......}|\\
\phantom{...}\ce{CH3\phantom{.}}\phantom{..}\ce{OH}\phantom{...}\ce{CH3}
\end{array}\]
Concept: undefined >> undefined
Advertisements
Write IUPAC name of the following compound:
\[\begin{array}{cc}
\ce{H3C - CH - CH2 - CH - CH - CH2 - CH3}\\
\phantom{}|\phantom{.............}|\phantom{......}|\phantom{.........}\\
\phantom{}\ce{OH}\phantom{..........}\ce{OH}\phantom{...}\ce{C2H5}\phantom{......}
\end{array}\]
Concept: undefined >> undefined
Write IUPAC name of the following compound:
\[\begin{array}{cc}
\ce{CH3 - CH - CH - CH3}\\
|\phantom{......}|\phantom{..}\\
\ce{OH}\phantom{...}\ce{OH}\phantom{}
\end{array}\]
Concept: undefined >> undefined
Write IUPAC name of the following compound:
\[\begin{array}{cc}
\ce{HO - CH2 - CH - CH2 - OH}\\
|\phantom{..}\\
\ce{OH}
\end{array}\]
Concept: undefined >> undefined
Write IUPAC name of the following compound:

Concept: undefined >> undefined
Write IUPAC name of the following compound:
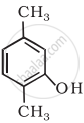
Concept: undefined >> undefined
Write IUPAC name of the following compound:

Concept: undefined >> undefined
Write IUPAC name of the following compound:
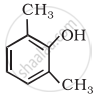
Concept: undefined >> undefined
Write IUPAC name of the following compound:
\[\begin{array}{cc}
\ce{CH3 - O - CH2 - CH - CH3}\\
\phantom{..........}|\\
\phantom{............}\ce{CH3}
\end{array}\]
Concept: undefined >> undefined
Write IUPAC name of the following compound:
C6H5 – O – C2H5
Concept: undefined >> undefined
Write IUPAC name of the following compound:
C6H5 – O – C7H15(n−)
Concept: undefined >> undefined
Write IUPAC name of the following compound:
\[\begin{array}{cc}
\ce{CH3 - CH2 - O - CH - CH2 - CH3}\\
\phantom{...}|\\
\phantom{.....}\ce{CH3}
\end{array}\]
Concept: undefined >> undefined
Write structures of the compounds whose IUPAC names are as follows:
3-Chloromethylpentan-1-ol.
Concept: undefined >> undefined
- Draw the structures of all isomeric alcohols of molecular formula C5H12O and give their IUPAC names.
- Classify the isomers of alcohols in the above question as primary, secondary and tertiary alcohols.
Concept: undefined >> undefined
Give IUPAC name of the following ether:
\[\begin{array}{cc}
\ce{C2H5OCH2 - CH - CH3}\\
\phantom{.....}|\\
\phantom{.......}\ce{CH3}
\end{array}\]
Concept: undefined >> undefined
Give IUPAC name of the following ether:
CH3OCH2CH2Cl
Concept: undefined >> undefined
Give IUPAC name of the following ether:
O2N – C6H4 – OCH3(p)
Concept: undefined >> undefined
Give IUPAC name of the following ether:
CH3CH2CH2OCH3
Concept: undefined >> undefined
Give IUPAC name of the following ether:

Concept: undefined >> undefined
